| Kidney Res Clin Pract > Epub ahead of print |
Abstract
Background
Methods
Results
Supplementary Materials
Notes
Data sharing statement
The data underlying this article are available on the homepage of Korea National Health and Nutrition Examination Survey (KNHANES), at https://knhanes.kdca.go.kr/knhanes/main.do. The datasets were derived from sources in the public domain: KNHANES 2008–2011, 2014–2018, https://knhanes.kdca.go.kr/knhanes/sub03/sub03_02_05.do.
Acknowledgments
Figure 1.
Selection of study population.
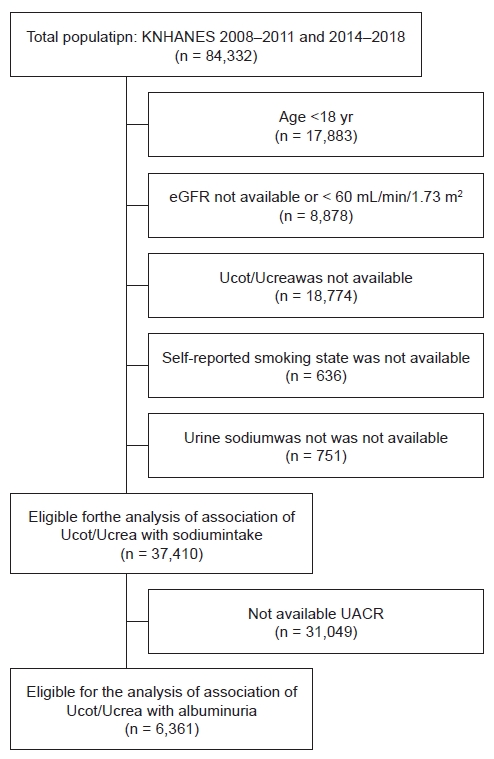
Figure 2.
Urine cotinine/creatinine ratio and cotinine concentration according to self-reported smoking status.
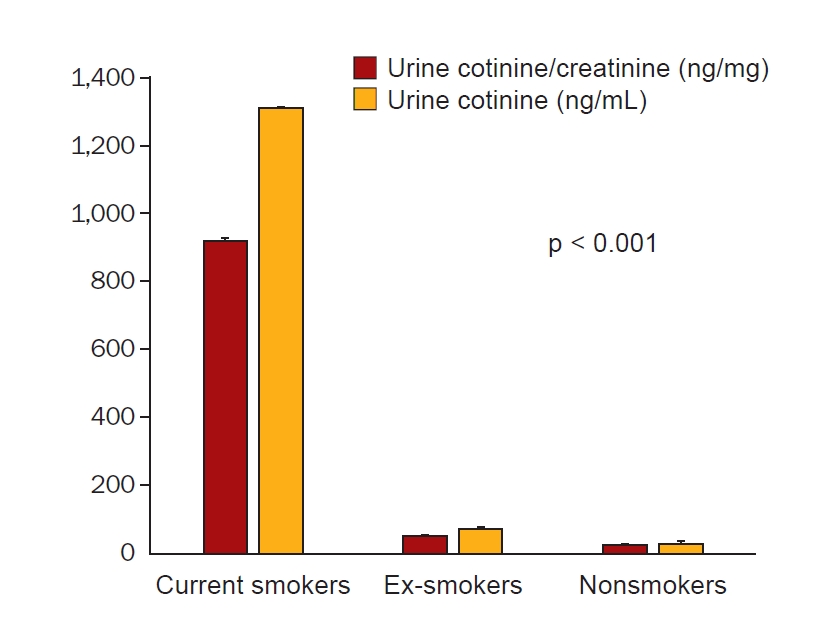
Table 1.
Data are expressed as number only, number (%), or mean ± standard deviation.
ALT, alanine aminotransferase; AST, aspartate transaminase; BMI, body mass index; BUN, blood urea nitrogen; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; LDL, low-density lipoprotein; SBP, systolic blood pressure; WBC, white blood cell.
Table 2.
Table 3.
| Ucot/Ucrea |
Model 1a |
Model 2b |
Model 3c |
|||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | |
| 1st tertile | ||||||
| 1st | Reference | Reference | Reference | |||
| 2nd | 0.641 (0.387–1.061) | 0.08 | 0.599 (0.359–0.999) | 0.049 | 0.688 (0.401–1.181) | 0.18 |
| 3rd | 1.220 (0.778–1.912) | 0.39 | 1.034 (0.653–1.638) | 0.89 | 1.131 (0.686–1.864) | 0.63 |
| 4th | 1.464 (0.914–2.343) | 0.11 | 1.097 (0.672–1.792) | 0.71 | 1.104 (0.643–1.896) | 0.72 |
| 2nd tertile | ||||||
| 1st | Reference | Reference | Reference | |||
| 2nd | 1.462 (0.787–2.717) | 0.23 | 1.312 (0.703–2.449) | 0.39 | 1.497 (0.778–2.882) | 0.23 |
| 3rd | 1.403 (0.770–2.556) | 0.27 | 1.247 (0.681–2.282) | 0.48 | 1.254 (0.660–2.384) | 0.49 |
| 4th | 2.181 (1.242–3.829) | 0.007 | 1.885 (1.060–3.353) | 0.03 | 1.647 (0.891–3.045) | 0.11 |
| 3rd tertile | ||||||
| 1st | Reference | Reference | Reference | |||
| 2nd | 1.500 (0.890–2.531) | 0.13 | 1.470 (0.866–2.496) | 0.15 | 1.715 (0.967–3.042) | 0.07 |
| 3rd | 1.710 (1.016–2.877) | 0.04 | 1.442 (0.848–2.454) | 0.18 | 1.411 (0.776–2.564) | 0.26 |
| 4th | 2.647 (1.648–4.251) | <0.001 | 2.325 (1.408–3.842) | 0.001 | 2.224 (1.262–3.916) | 0.006 |
References
- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 801 View
- 22 Download
- ORCID iDs
-
Young-Bin Son

https://orcid.org/0000-0003-0009-5866Tae-Bum Kim

https://orcid.org/0000-0001-9300-6732Hyeon-Jin Min

https://orcid.org/0000-0003-4377-4172Jihyun Yang

https://orcid.org/0000-0002-8616-238XMyung-Gyu Kim

https://orcid.org/0000-0001-8807-2452Sang Kyung Jo

https://orcid.org/0000-0002-0496-0258Won Yong Cho

https://orcid.org/0000-0001-8627-8940Se Won Oh

https://orcid.org/0000-0003-3795-9322 - Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement table 1
Supplement table 1 Print
Print















