| Kidney Res Clin Pract > Epub ahead of print |
Abstract
Background
Methods
Results
Notes
Additional information
The approval numbers of each Institutional Review Board are as follows: Seoul National University Hospital (No. 1810-132-982), Seoul National University Bundang Hospital (No. B-1901-514-409), Severance Hospital (No. 4-2019-0152), Kangbuk Samsung Hospital (No. 2019-04-011), The Catholic University of Korea, Seoul St. MaryŌĆÖs Hospital (No. KC19OEDI0263), Gachon University Gil Medical Center (No. GAIRB2019-154), Nowon Eulji Medical Center, Eulji University (No. 201904-01), Chonnam National University Hospital (No. 2021-07-111), Inje University Pusan Paik Hospital (No. 2018-01-203), Hallym University Dongtan Sacred Heart Hospital (No. 2019-11-006), National Health Insurance Service Ilsan Hospital (No. 2019-12-008), Seoul National University Boramae Medical Center (No. 20-2019-76), Pusan National University Hospital (No. 1912-019-086), Chungnam National University Hospital (No. 2021-07-111).
Conflicts of interest
Tae-Hyun Yoo is the Editor-in-Chief of Kidney Research and Clinical Practice and was not involved in the review process of this article. All authors have no other potential conflicts of interest relevant to this article.
Funding
This work was supported by the Research Program funded by the Korea Disease Control and Prevention Agency (2011E3300300, 2012E3301100, 2013E3301600, 2013E3301601, 2013E3301602, 2016E3300200, 2016E3300201, 2016E3300202, 2019E320100, 2019E320101, 2019E320102, and 2022-11-007). The funding sources had no role in the design and conduct of the study, collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Data sharing statement
The data presented in this study are available on request from the corresponding author.
AuthorsŌĆÖ contributions
Conceptualization, Investigation: YEK, THY
Methodology: YEK, HWK, JTP, SHH, SWK, THY
Data curation: HWK, THY
Formal analysis: YEK, HWK, JTP, SHH, SWK, THY
Supervision: THY, HWK, JTP, SHH, SWK, SS, KBL, JL, KHO
WritingŌĆōoriginal draft: YEK, THY
WritingŌĆōreview & editing: YEK, THY
All authors critically reviewed and approved the final manuscript.
Acknowledgments
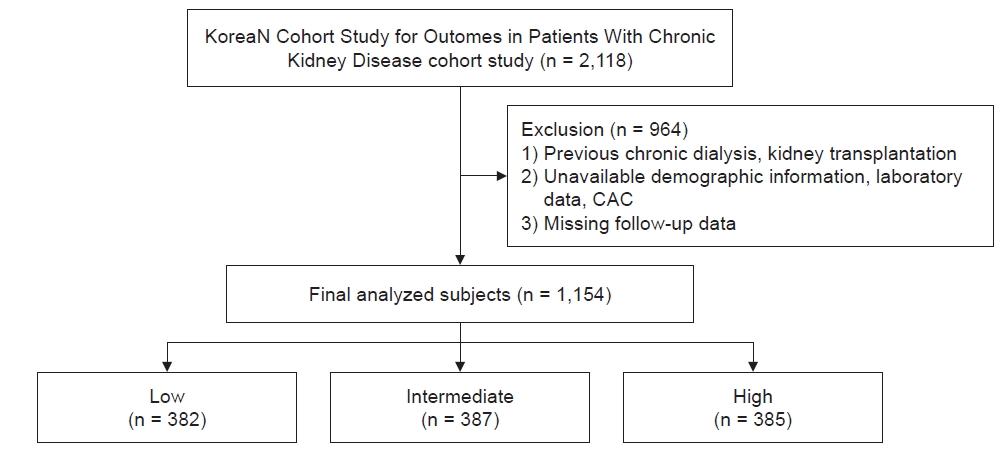
Figure┬Ā1.
Flow diagram for the patients enrolled in this study.
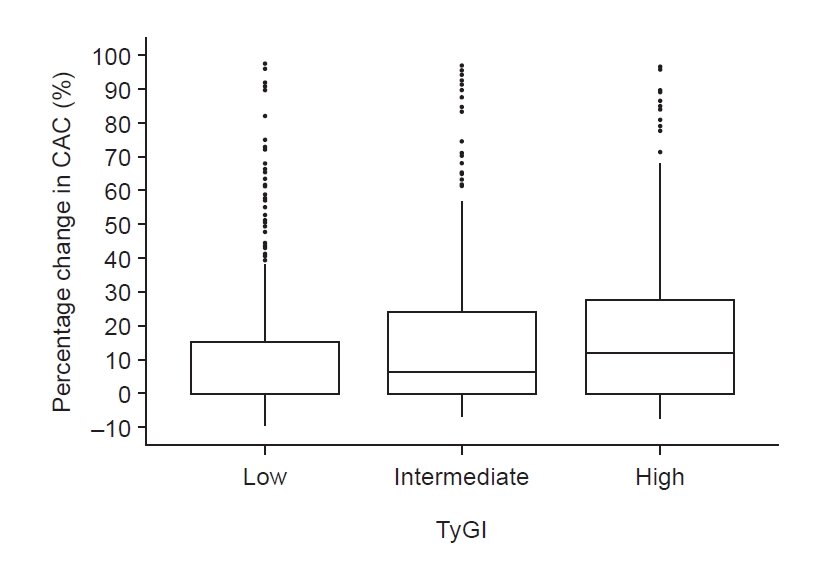
Figure┬Ā2.
Percentage change in CAC among different TyGI groups.
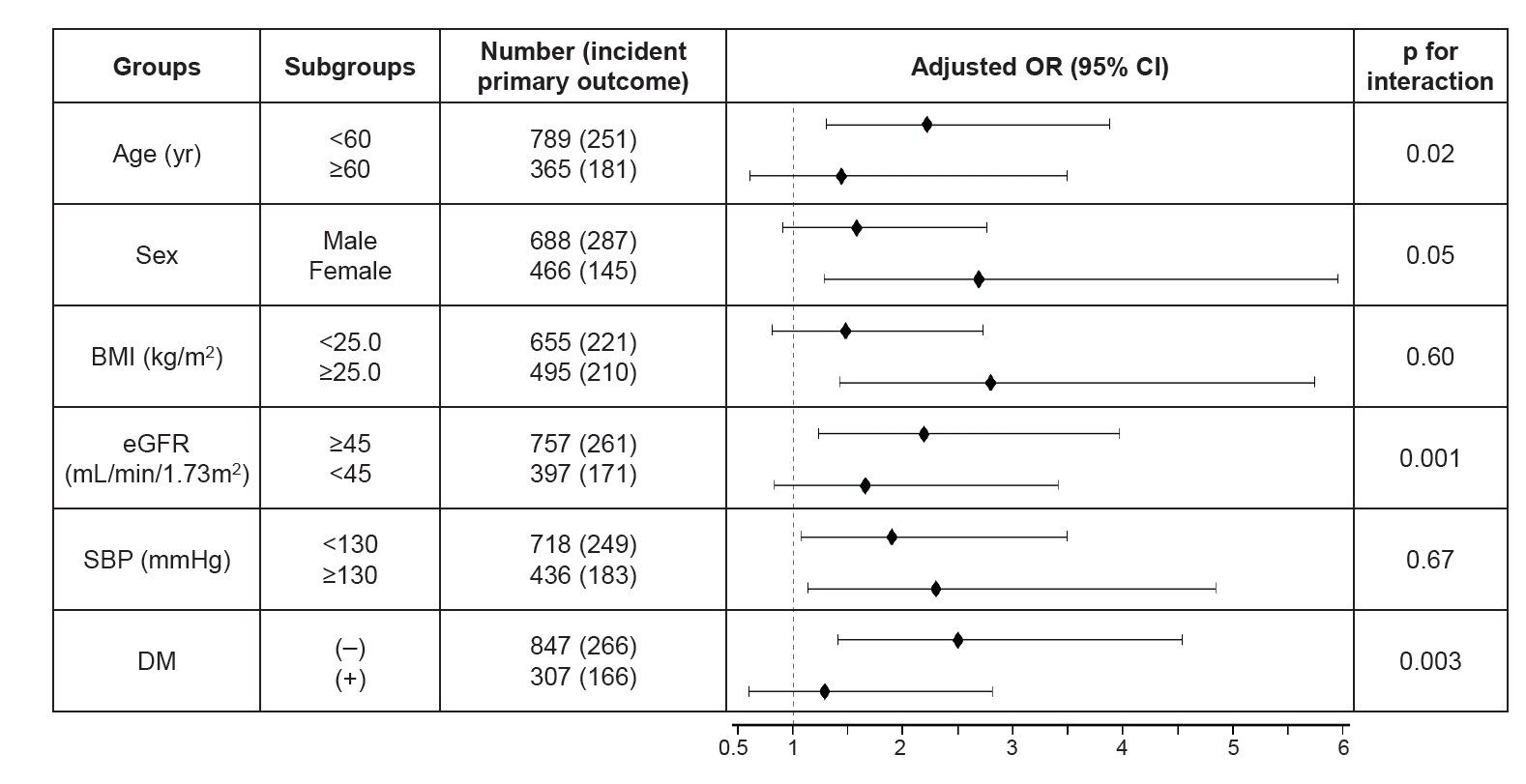
Figure┬Ā3.
Forest plot for modification effects of subgroups on the relationship between triglyceride-glucose index and CAC progression.
Table┬Ā1.
Data are expressed as number (%) or mean ┬▒ standard deviation.
DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; FGF-23, fibroblast growth factor 23; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PTH, parathyroid hormone; SBP, systolic blood pressure; TyGI, triglyceride-glucose index.
Table┬Ā2.
Table┬Ā3.
| Variable |
Model 1a |
Model 2b |
Model 3c |
|||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | |
| TyGI | ||||||
| ŌĆāLow | Reference | - | Reference | - | Reference | - |
| ŌĆāIntermediate | 1.50 (1.11ŌĆō2.03) | 0.009 | 1.22 (0.84ŌĆō1.77) | 0.31 | 1.49 (0.86ŌĆō2.56) | 0.16 |
| ŌĆāHigh | 2.15 (1.59ŌĆō2.91) | <0.001 | 1.79 (1.22ŌĆō2.63) | 0.003 | 2.11 (1.14ŌĆō3.88) | 0.02 |
| TyGI (per 1-point increase) | 1.39 (1.08ŌĆō1.79) | 0.01 | 1.68 (1.30ŌĆō2.17) | <0.001 | 1.55 (1.06ŌĆō1.76) | 0.003 |
Table┬Ā4.
| Variable |
Model 1a |
Model 2b |
Model 3c |
|||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | |
| TyGI | ||||||
| ŌĆāLow | Reference | - | Reference | - | Reference | - |
| ŌĆāIntermediate | 1.82 (1.34ŌĆō2.48) | <0.001 | 1.48 (1.00ŌĆō2.21) | 0.05 | 1.57 (0.86ŌĆō2.90) | 0.15 |
| ŌĆāHigh | 2.29 (1.69ŌĆō3.11) | <0.001 | 1.67 (1.11ŌĆō2.51) | 0.01 | 1.85 (0.94ŌĆō3.68) | 0.08 |
| TyGI (per 1-point increase) | 1.82 (1.50ŌĆō2.22) | <0.001 | 1.53 (1.17ŌĆō2.00) | 0.002 | 1.61 (1.01ŌĆō2.60) | 0.048 |
References
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 169 View
- 5 Download
- ORCID iDs
-
Ye Eun Ko

https://orcid.org/0009-0002-5369-2031Hyung Woo Kim

https://orcid.org/0000-0002-6305-452XJung Tak Park

https://orcid.org/0000-0002-2325-8982Seung Hyeok Han

https://orcid.org/0000-0001-7923-5635Shin-Wook Kang

https://orcid.org/0000-0002-5677-4756Suah Sung

https://orcid.org/0000-0002-7100-9964Kyu-Beck Lee

https://orcid.org/0000-0002-3904-5404Joongyub Lee

https://orcid.org/0000-0003-2784-3772Kook-Hwan Oh

https://orcid.org/0000-0001-9525-2179Tae-Hyun Yoo

https://orcid.org/0000-0002-9183-4507 - Related articles
-
Clinical Features of Ischemic Colitis in Patients with Chronic Kidney Disease2009 December;28(6)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print















