| Kidney Res Clin Pract > Volume 41(6); 2022 > Article |
|
Abstract
Background
Methods
Results
Notes
Authors’ contributions
Conceptualization: HRJ
Data curation: JJ, KP, HSL
Formal analysis: JJ, KP, KK
Investigation: JJ, KP, JEL, KK, WH, YGK, DJK, HRJ
Methodology: JJ, HRJ
Project administration: KWL, JEL, JBP, WH, YGK, DJK
Visualization: JJ, KP
Writing–original draft: JJ, KP, HSL, KWL
Writing–review & editing: JJ, KP, HRJ, JEL, JBP, WH, YGK, DJK
All authors read and approved the final manuscript.
Acknowledgments
Supplementary Materials
Figure 1.
Serial follow-up measurements of postoperative proteinuria and eGFR.
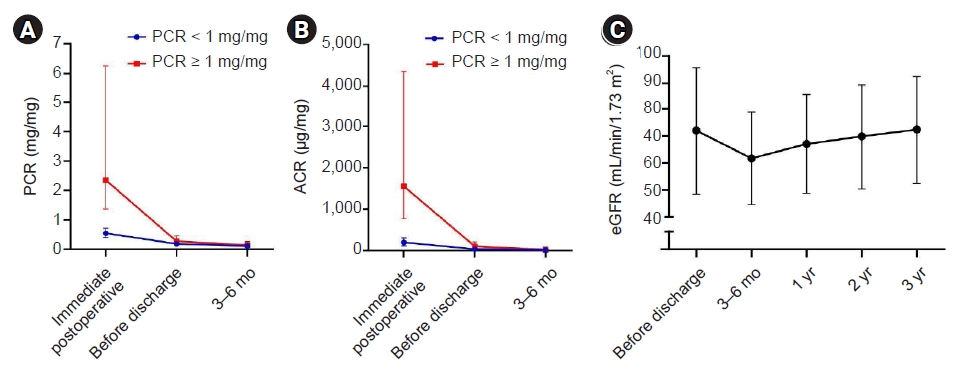
Figure 2.
Correlations between spot urine PCR or ACR and 24-hour urine protein excretion.

Figure 3.
Serial follow-up measurements (for up to 3 years) of the postoperative eGFR according to the degree of proteinuria at postoperative 3 to 6 months after KT.

Table 1.
| Characteristic | Total | Low-PCR groupa | High-PCR groupb | p-value |
|---|---|---|---|---|
| No. of patients | 353 | 195 | 158 | |
| Age (yr) | 51 (43–59) | 48 (41–56) | 55 (46–61) | <0.001 |
| Male sex | 222 (62.9) | 119 (61.0) | 103 (65.2) | 0.44 |
| RRT modality before KT | 0.17 | |||
| Hemodialysis | 265 (75.1) | 139 (71.3) | 126 (79.7) | |
| Peritoneal dialysis | 30 (8.5) | 20 (10.3) | 10 (6.3) | |
| No dialysis | 58 (16.4) | 36 (18.5) | 22 (13.9) | |
| Cause of ESRD | 0.61 | |||
| GN | 97 (27.5) | 54 (27.7) | 43 (27.2) | |
| Diabetes mellitus | 112 (31.7) | 50 (25.6) | 62 (39.2) | |
| Hypertension | 33 (9.3) | 24 (12.3) | 9 (5.7) | |
| Cystic kidney disease | 16 (4.5) | 9 (4.6) | 7 (4.4) | |
| Others | 40 (11.3) | 25 (12.8) | 15 (9.5) | |
| Unknown | 55 (15.6) | 33 (16.9) | 22 (13.9) | |
| BMI (kg/m2) | 22.7 (20.7–25.1) | 22.7 (20.6–25.5) | 22.6 (20.6–24.5) | 0.34 |
| Diabetes mellitus | 114 (32.3) | 51 (26.2) | 63 (39.9) | 0.008 |
| Cardiovascular disease | 27 (7.6) | 12 (6.2) | 15 (9.5) | 0.314 |
| SBP (mmHg) | 141 (128–153) | 138 (125–150) | 145 (134–157) | 0.001 |
| DBP (mmHg) | 83 (74–90) | 83 (75–90) | 83 (74–91) | 0.88 |
| Donor age (yr) | 50 (38–60) | 48 (37–56) | 52 (43–64) | <0.001 |
| Male donor | 205 (58.1) | 107 (54.9) | 98 (62.0) | 0.19 |
| Donor height (cm) | 167 (159–172) | 165 (158–172) | 167 (160–173) | 0.26 |
| Donor weight (kg) | 66.0 (57.9–74.3) | 66.2 (57.8–74.4) | 66.0 (57.9–74.3) | 0.90 |
| Donor BMI (kg/m2) | 24.1 (21.7–26.3) | 24.5 (22.1–26.3) | 23.9 (21.5–26.3) | 0.34 |
| Donor serum creatinine (mg/dL) | 0.89 (0.70–1.24) | 0.81 (0.67–0.95) | 1.14 (0.84–1.87) | <0.001 |
| Donor status | <0.001 | |||
| Living | 186 (52.7) | 146 (74.9) | 40 (25.3) | |
| Deceased | 167 (47.3) | 49 (25.1) | 118 (74.7) | |
| ABO incompatibility | 93 (26.3) | 66 (33.8) | 27 (17.1) | <0.001 |
| Donor-specific antibody | 56 (15.8) | 26 (13.5) | 20 (12.9) | 0.875 |
| Induction therapy | <0.001 | |||
| No induction | 1 (0.3) | 1 (0.5) | 0 (0) | |
| ATG | 218 (61.8) | 104 (53.3) | 114 (72.6) | |
| ATG + rituximab | 73 (20.7) | 50 (25.6) | 23 (14.6) | |
| Basiliximab | 60 (17.0) | 40 (20.5) | 20 (12.7) | |
| Basiliximab + rituximab | 1 (0.3) | 0 (0) | 1 (0.6) | |
| Delayed graft function | 39 (11.0) | 4 (2.1) | 35 (22.2) | <0.001 |
| Acute rejection | 129 (36.5) | 79 (40.5) | 50 (31.6) | 0.10 |
| Antibody-mediated rejection | 25 (7.1) | 17 (8.7) | 8 (5.1) | 0.18 |
| BK virus nephropathy | 21 (5.9) | 11 (5.6) | 10 (6.3) | 0.79 |
| Post-KT glomerular pathology | 56 (15.9) | 26 (13.3) | 30 (19.0) | 0.41 |
| Focal segmental glomerulosclerosis | 6 (1.7) | 3 (1.5) | 3 (1.9) | |
| IgA nephropathy | 7 (2.0) | 4 (2.1) | 3 (1.9) | |
| Membranoproliferative GN | 2 (0.6) | 2 (1.0) | 0 (0) | |
| Diabetic nephropathy | 9 (2.5) | 1 (0.5) | 8 (5.1) | |
| CNI toxicity | 13 (3.7) | 7 (3.6) | 6 (3.8) | |
| Others | 19 (5.4) | 9 (4.6) | 10 (6.3) | |
| Interval from KT to diagnosis of glomerular pathology (day) | 193 (145–408) | 362 (14–414) | 86 (15–398) | 0.565 |
| Treatment of ACEi/ARB within 1 year after KT | 34 (9.6) | 16 (8.2) | 18 (11.4) | 0.313 |
Data are expressed as number only, median (interquartile range), and number (%).
ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; ATG, antithymocyte globulin; BMI, body mass index; CNI, calcineurin inhibitor; DBP, diastolic blood pressure; ESRD, end-stage renal disease; GN, glomerulonephritis; IgA, immunoglobulin A; KT, kidney transplantation; PCR, spot urine protein-to-creatinine ratio; RRT, renal replacement therapy; SBP, systolic blood pressure.
Table 2.
| Variable |
Unadjusted |
Adjusteda |
||
|---|---|---|---|---|
| β coefficient | p-value | β coefficient | p-value | |
| PCR, immediately postoperation | –3.68 | 0.03 | 2.88 | 0.18 |
| ACR, immediately postoperation | –3.53 | 0.01 | 1.72 | 0.33 |
| PCR, before discharge | –4.73 | 0.18 | –2.62 | 0.46 |
| ACR, before discharge | –4.45 | 0.02 | –2.39 | 0.23 |
| PCR, 3–6 months posttransplant | –11.51 | <0.001 | –7.97 | 0.02 |
| ACR, 3–6 months posttransplant | –8.62 | <0.001 | –6.68 | 0.001 |
Table 3.
References
- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 2,610 View
- 80 Download
- ORCID iDs
-
Junseok Jun

https://orcid.org/0000-0003-2532-0177Kyungho Park

https://orcid.org/0000-0002-4888-7523Hyun Suk Lee

https://orcid.org/0000-0003-4444-0537Kyo Won Lee

https://orcid.org/0000-0002-2722-7817Jung Eun Lee

https://orcid.org/0000-0002-4387-5291Jae Berm Park

https://orcid.org/0000-0001-9117-2278Kyunga Kim

https://orcid.org/0000-0002-0865-2236Wooseong Huh

https://orcid.org/0000-0001-8174-5028Yoon-Goo Kim

https://orcid.org/0000-0002-8176-295XDae Joong Kim

https://orcid.org/0000-0001-7526-1107Hye Ryoun Jang

https://orcid.org/0000-0001-9856-6341 - Related articles
-
The effect of periodontitis on recipient outcomes after kidney transplantation2022 January;41(1)
A Case of Invasive Fungal Sinusitis after Kidney Transplantation2009 July;28(4)




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement 1
Supplement 1 Print
Print















