| Kidney Res Clin Pract > Volume 41(2); 2022 > Article |
|
Abstract
Background
Methods
Results
Notes
AuthorsŌĆÖ contributions
Conceptualization: SK
Study design: JYR, EB
Data curation, Formal analysis: HES, SSH, SK
Investigation: SSH, JJM, JP, HES
Supervision: SK, SSH, JCJ, HJC, KYN, DC
WritingŌĆōoriginal draft: HES
WritingŌĆōreview & editing: HES, SSH, SK
All authors read and approved the final manuscript.
Figure┬Ā1.
Kaplan-Meier curves for death by groups, based on the presence of AKI or AHF.

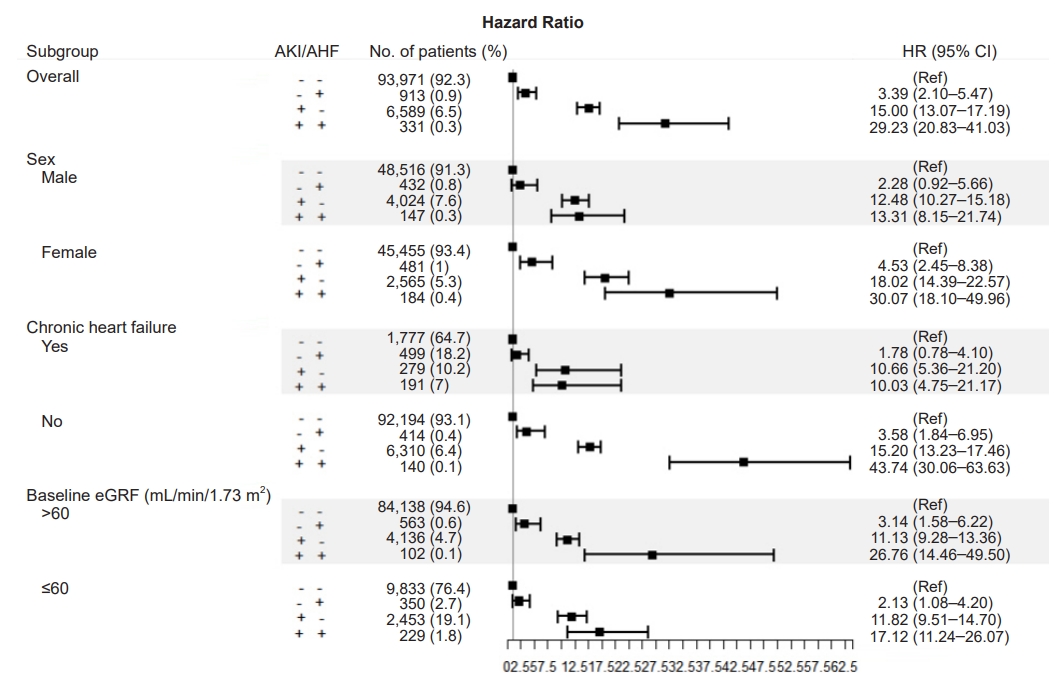
Figure┬Ā2.
HRs of death within 1 month in subgroups based on sex, underlying chronic heart disease, and baseline eGFR.
Figure┬Ā3.
HRs of variables included in the multivariable Cox regression model for death within 1 month.

Table┬Ā1.
Data are expressed as number only, number (%), mean ┬▒ standard deviation, or median (interquartile range).
Operations were classified into two categories by expected surgery time: major operation defined as surgery duration Ōēź 1 hour, and minor operation defined as surgery duration < 1 hour.
AHF, acute heart failure; AKI, acute kidney injury; BMI, body mass index; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; ICU, intensive care unit; NSAID, nonsteroidal anti-inflammatory drug; RAS, renin-angiotensin system; WBC, white blood cell.
Table┬Ā2.
RERI (95% CI), 11.846 (2.426ŌĆÆ21.266); p = 0.014. AP (95% CI), 0.405 (0.211ŌĆÆ0.600); p < 0.001. SI (95% CI), 1.723 (1.227ŌĆÆ2.418); p = 0.002.
RERI and AP were >0, and SI was >1, suggesting an additive interaction between AKI and AHF. Hazard ratio was adjusted for age over 75 years, sex, body mass index, admission duration in days, comorbidities (hypertension, diabetes mellitus, chronic kidney disease, chronic heart failure, ischemic heart disease, liver disease, cerebrovascular disease, chronic obstructive pulmonary disease, and cancer), and medications used in the past 6 months just before admission (diuretics, renin-angiotensin system blockers, beta blockers, calcium-channel blockers, nonsteroidal anti-inflammatory drugs, vancomycin, and vasopressors).
AP, attributable proportion due to interaction; AKI, acute kidney injury; AHF, acute heart failure; CI, confidence interval; HR, hazard ratio; RERI, relative excess risk of interaction; SI, synergy index.
Table┬Ā3.
Relative excess risk of interaction (95% CI), 47.042 (11.519ŌĆō82.564); p = 0.009. Attributable proportion due to interaction (95% CI), 0.668 (0.496ŌĆō0.840); p < 0.001.
Synergy index (95% CI), 3.11 (1.83ŌĆÆ5.26); p < 0.001.
Hazard ratio was adjusted for sex, body mass index, admission days, comorbidities (hypertension, diabetes mellitus, chronic kidney disease, chronic heart failure, ischemic heart disease, liver disease, cerebrovascular disease, chronic obstructive pulmonary disease, and cancer), and medications used in the last 6 months just before admission (diuretics, renin-angiotensin system blockers, beta blockers, calcium-channel blockers, nonsteroidal anti-inflammatory drugs, vancomycin, and vasopressors).
AKI, acute kidney injury; AHF, acute heart failure; CI, confidence interval; HR, hazard ratio.
Table┬Ā4.
RERI (95% CI), 2.428 (ŌĆō3.626 to 8.482); p = 0.432. AP (95% CI), 0.170 (ŌĆō0.187 to 0.526); p = 0.350. SI (95% CI), 1.224 (0.770ŌĆÆ1.943); p = 0.393.
RERI and AP were > 0 and SI was > 1 in patients who were Ōēż75 years old, while this was not the case in patients over 75 years old. AKI and AHF individually increased mortality within 1 month, regardless of age.
Hazard ratio was adjusted for sex, body mass index, admission days, comorbidities (hypertension, diabetes mellitus, chronic kidney disease, chronic heart failure, ischemic heart disease, liver disease, cerebrovascular disease, chronic obstructive pulmonary disease, and cancer), and medications used in the last 6 months just before admission (diuretics, renin-angiotensin system blockers, beta blockers, calcium-channel blockers, nonsteroidal anti-inflammatory drugs, vancomycin, and vasopressors).
AP, attributable proportion due to interaction; AKI, acute kidney injury; AHF, acute heart failure; CI, confidence interval; HR, hazard ratio; RERI, relative excess risk of interaction; SI, synergy index.
References
-
METRICS

- ORCID iDs
-
Hyung Eun Son

https://orcid.org/0000-0002-8719-3823Jong Joo Moon

https://orcid.org/0000-0003-1034-0837Jeong-min Park

https://orcid.org/0000-0003-3493-8235Ji Young Ryu

https://orcid.org/0000-0003-4134-1007Eunji Baek

https://orcid.org/0000-0001-9226-7703Jong Cheol Jeong

https://orcid.org/0000-0003-0301-7644Ho Jun Chin

https://orcid.org/0000-0002-3710-0190Ki Young Na

https://orcid.org/0000-0002-8872-8236Dong-wan Chae

https://orcid.org/0000-0001-9401-892XSeung Seok Han

https://orcid.org/0000-0003-0137-5261Sejoong Kim

https://orcid.org/0000-0002-7238-9962 - Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print















