Acute kidney injury in patients with acute-on-chronic liver failure: clinical significance and management
Article information
Abstract
Acute-on-chronic-liver failure (ACLF) refers to a phenomenon in which patients with chronic liver disease develop multiple organ failure due to acute exacerbation of underlying liver disease. More than 10 definitions of ACLF are extant around the world, and there is lack of consensus on whether extrahepatic organ failure is a main component or a consequence of ACLF. Asian and European consortiums have their own definitions of ACLF. The Asian Pacific Association for the Study of the Liver ACLF Research Consortium does not consider kidney failure as a diagnostic criterion for ACLF. Meanwhile, the European Association for the Study of the Liver Chronic Liver Failure and the North American Consortium for the Study of End-stage Liver Disease do consider kidney failure as an important factor in diagnosing and assessing the severity of ACLF. When kidney failure occurs in ACLF patients, treatment varies depending on the presence and stage of acute kidney injury (AKI). In general, the diagnosis of AKI in cirrhotic patients is based on the International Club of Ascites criteria: an increase of 0.3 mg/dL or more within 48 hours or a serum creatinine increase of 50% or more within one week. This study underscores the importance of kidney failure or AKI in patients with ACLF by reviewing its pathophysiology, prevention methods, and treatment approaches.
Introduction
Broadly, there are three clinical stages of liver cirrhosis; compensated cirrhosis, decompensated cirrhosis, and late decompensated cirrhosis (DC) [1]. However, in 1997, Jalan et al. [2] reported several cases of young cirrhotic patients who died shortly after a transjugular intrahepatic portosystemic shunt procedure for variceal bleeding due to increased intracranial pressure and rapid liver function deterioration. It is inappropriate to classify these cases into the extant cirrhosis stages because increased intracranial pressure is frequently found in acute liver failure rather than in DC. Accordingly, acute exacerbation of existing chronic liver disease has been officially referred to as acute-on-chronic-liver failure (ACLF) [3].
As suggested, ACLF refers to a specific condition of preexisting chronic liver disease (with or without liver cirrhosis) that is associated with rapid deterioration of liver function, multiorgan failure, and increased mortality [4–9]. The prognosis of ACLF is notably different from that of cirrhosis in that ACLF tends to get worse suddenly [4,10]. The socioeconomic burden of ACLF is estimated to be much larger than that of liver cirrhosis. In the United States, significantly greater per capita hospitalization costs ($51,000 vs. $14,000), hospitalization days (16 days vs. 7 days), and mortality rates (50% vs. 7%) were reported in ACLF patients than in liver cirrhosis patients [11]. ACLF is comparable to acute kidney injury (AKI) in chronic kidney disease (CKD).
Various definitions of acute-on-chronic-liver failure
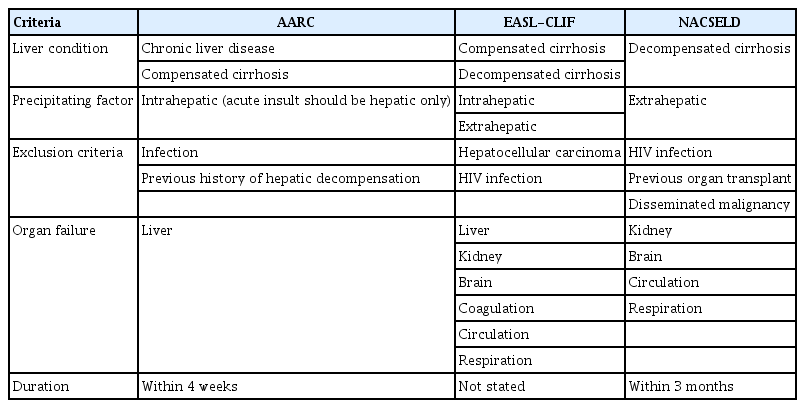
Within the field of hepatology, ACLF has been extensively studied, and more than 1,000 papers have been published [9]. The classification and prognosis of ACLF vary across countries, race and ethnic groups, and etiologies, reflecting inconsistent views, and there are more than 10 definitions of ACLF around the world. The most widely accepted clinical definitions are those outlined by the Asian Pacific Association for the Study of the Liver ACLF Research Consortium (AARC) [12], the European Association for the Study of the Liver Chronic Liver Failure (EASL-CLIF) [13], and the North American Consortium for the Study of End-stage Liver disease (NACSELD) [14]. They are consistent in defining ACLF as a high-mortality disease that involves acute exacerbation of existing liver disease. However, their diagnostic considerations differ in terms of 1) liver condition, 2) organ failures, and 3) precipitating factors. Table 1 presents the most representative diagnostic criteria of the AARC, the EASL-CLIF, and the NACSELD [12–14].
Multiple perspectives on organ failure and the importance of kidney failure
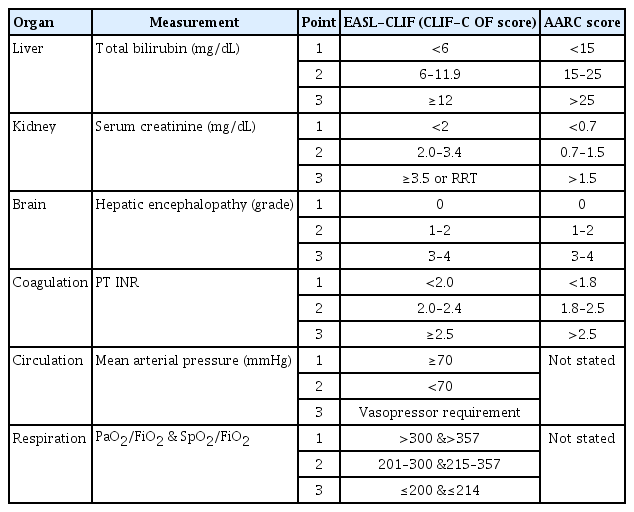
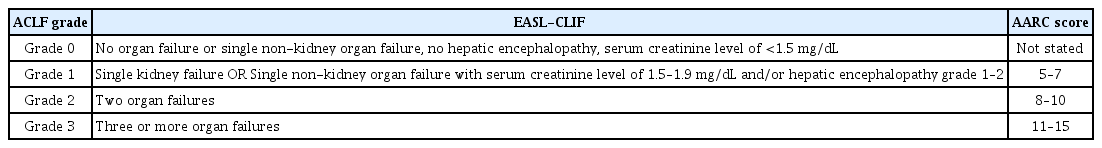
As shown in Table 1, the AARC considers extrahepatic organ failure a consequence of ACLF and does not include it in the diagnostic criteria. The EASL-CLIF and the NACSELD, however, incorporate extrahepatic organ failure as an important component of the diagnostic criteria for ACLF. To evaluate organ failure, the AARC, the EASL-CLIF, and the NACSELD respectively consider one, five, and four organs (Table 1). The definitions of organ failure of the EASL-CLIF and the AARC are presented in Table 2 [5–7]. The ACLF grades are classified depending on the number of organ failures, which is directly related to mortality (Table 3). With a single organ failure, the 28-day mortality rate of ACLF was 6.3%, whereas that for failure of four or more organs reached 88.9% [6,15]. Kidney is known to be easily affected by extrahepatic organ failure in ACLF patients, and the incidence of kidney failure (55.8%) was higher than that of liver failure (43.6%) in the CANONIC study [15,16]. Also, kidney failure can more adversely affect the prognosis of ACLF than can failure of any other single organ [17–19]. In non-kidney organ failure, the 28-day mortality rate of ACLF patients was 6.3%, while that of patients with kidney failure alone reached as high as 18.6% [15]. Therefore, failure of a single organ other than the kidney is considered ACLF grade 0, whereas failure of the kidney alone is considered ACLF grade 1 (Table 3).
Definitions of acute kidney injury and hepatorenal syndrome in patients with cirrhosis
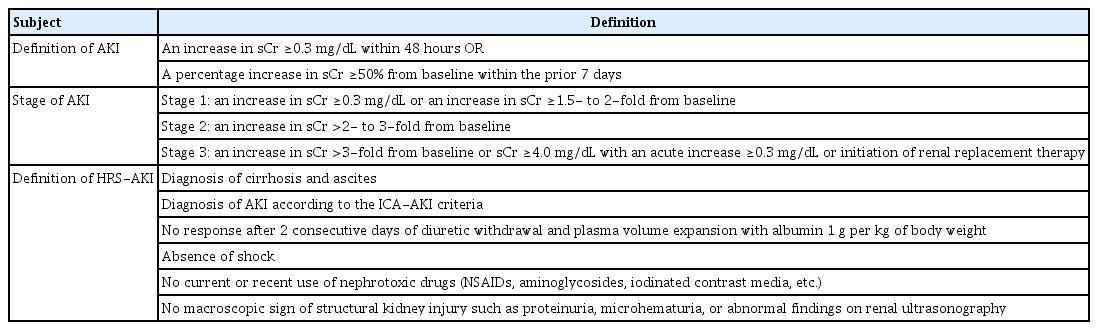
For diagnosis of AKI in cirrhosis patients, the definition of the International Club of Ascites (ICA) and KDIGO (Kidney Disease: Improving Global Outcomes) AKI guidelines have been most widely used [20,21]. In 2007, the ICA introduced the concepts of hepatorenal syndrome (HRS)-1 and -2, characterized by rapid deterioration and refractory ascites, respectively [22]; the concepts of AKI and HRS were updated in 2015 [20]. AKI was defined as serum creatinine level of 1.5 mg/dL or greater or a serum creatinine increase of 50% or greater compared to the baseline value [22]. However, AKI was redefined as a serum creatinine increase by 0.3 mg/dL or greater within 48 hours or a serum creatinine increase by 50% or greater compared to the baseline value within 1 week. This was an important change that eliminated the absolute standard of 1.5 mg/dL of serum creatinine level in the diagnosis of AKI. Table 4 summarizes the most commonly used diagnostic standards for HRS and guidelines for determining AKI stages (adapted from the ICA definition) [20]. Due to poor nutritional status and low muscle mass in cirrhotic patients, there have been persistent concerns that the creatinine-based glomerular filtration rate (GFR) formula might overestimate actual kidney function [23]. In particular, overestimation of renal function with the creatinine-based GFR formula reached up to about 50% in patients with severely impaired liver function [24]. Thus, clinicians should pay attention to deterioration of renal function and start treatment for AKI in a timely manner, well before serum creatinine level increases and reaches 1.5 mg/dL. Studies have shown that high AKI grade is associated with poor prognosis in cirrhotic patients. Even in cirrhotic patients with AKI grade I, 3-month survival rate was reduced to 84% [25–27].
Meanwhile, as the definition of AKI in cirrhosis patients was updated and the absolute value of serum creatinine is no longer involved, HRS-1, which was previously determined by an increase of serum creatinine level 2.5 mg/dL or greater, was renamed HRS-AKI [20]. These changes are primarily based on the low response rate of patients with high serum creatinine to treatment with terlipressin plus albumin for HRS [28–30]. The newly revised definition recommends starting treatment with vasoconstrictor and albumin in the early stage of AKI, even if serum creatinine level is less than 2.5 mg/dL. The level of serum creatinine tends to overestimate renal function in cirrhotic patients and varies greatly by day. Hence, absolute standards such as serum creatinine level of 1.5 mg/dL (AKI) or 2.5 mg/dL (HRS) should be avoided, and clinicians need to start treating AKI and HRS as early as possible in consideration of the dynamic fluctuation present in serum creatinine level.
In the same vein, attempts have been made to rename HRS type 2, which is characterized by a slow increase of serum creatinine and refractory ascites, to HRS-non-AKI (NAKI) and to have it be divided into HRS-acute kidney disease and HRS-CKD depending on the rate of exacerbation [31]. However, HRS-NAKI is rarely reported and analyzed in ACLF patients, so we will not discuss it in detail in this review.
Prevalence of acute kidney injury in patients with acute-on-chronic-liver failure
According to the CANONIC study, the prevalence of AKI in ACLF patients is 69% [15]. Among them, the proportions of kidney dysfunction (serum creatinine level of 1.5–1.9 mg/dL) and kidney failure (serum creatinine level of >2 mg/dL) cases were respectively 19.1% and 80.9% [15]. These reports were made prior to revision of the ICA-AKI standard in 2015. Thus, the values are likely to be higher if the current standards of AKI are applied. Even in the ACLF patient group based on the AARC criteria, the prevalence of AKI was estimated to be around 22.8% to 51%, much higher than that (20%) in cirrhotic patients without ACLF [12]. In research conducted in South Korea, 340 among 1,470 patients (23.1%) hospitalized for chronic liver disease had ACLF, and 49% of ACLF patients had kidney failure [32].
Differences in characteristics of acute kidney injury between acute-on-chronic-liver failure and decompensated cirrhosis
In the past, the concept of ACLF was not well established, and there was a lack of distinction between cirrhotic patients with DC and those with ACLF in terms of reported prognosis and clinical outcome. Accumulating evidence suggests that the pattern of organ failure between ACLF and DC is different [33]. Even though the incident rate of AKI was not significantly different between ACLF and DC patient groups (around 13%–25%), the phenotype of AKI was notably different [34,35]. The DC-AKI group tended to be functional and volume-responsive, while the ACLF-AKI group had more severe structural damage [33]. In addition, the probability of AKI progression was relatively low in the DC-AKI group, and resolution was rare [33]. On the other hand, the ACLF-AKI group had a high possibility of AKI progression, as well as of resolution [33,34]. The ACLF-AKI group required renal replacement therapy (RRT) more frequently and had lower response rate to terlipressin than the DC-AKI group, possibly due to severe and extensive systemic inflammation. Table 5 summarizes the differences in characteristics of the ACLF-AKI and DC-AKI groups. However, more studies are needed to better understand the difference between DC-AKI and ACLF-AKI groups.
A prior study on the difference between DC and ACLF patient groups suggests that they have different metabolic profiles such that ACLF patients showed increased skeletal muscle catabolism resulting in release of amino acid to increase nonessential amino acid/glucose and methionine compared with DC patients [36]. Also, the level of spermidine, an inducer of anti-inflammatory autophagy, was decreased in ACLF patients [36].
Another difference between DC and ACLF patient groups is the gut microbiome. Progression of cirrhosis was associated with a reduction of gene and metagenomic species richness in the gut, with maximal changes in ACLF patients [37].
Precipitating factors
Common causes of AKI in general cirrhotic patients include use of diuretics, gastrointestinal bleeding, large volume paracentesis, infection, and use of nephrotoxic drugs such as nonsteroidal anti-inflammatory drugs (NSAIDs) [38–40]. The precipitating factors of AKI and ACLF are similar. ACLF can be precipitated by both hepatic and nonhepatic factors [41,42]. In the AARC criteria, acute exacerbation caused by nonhepatic precipitating factors such as infection and gastrointestinal bleeding is excluded from the ACLF definition. However, both nonhepatic and hepatic precipitating factors are considered in the EASL-CLIF criteria. According to studies conducted in Western countries (CANONIC study, EASL-CLIF cohort), the most important causes of acute exacerbation were active alcohol drinking and bacterial infection [15]. On the other hand, according to studies conducted in Eastern countries, hepatitis B virus reactivation, hepatitis A or E virus infection, active alcohol drinking, and bacterial infection were major precipitating factors [43,44]. In South Korean ACLF patients, the most common precipitating factor was active alcohol drinking (41%), followed by gastrointestinal bleeding (31%) and bacterial infection (18.5%) [32]. However, the cause of acute exacerbation in about 40% to 50% of ACLF patients is not clearly elucidated in many studies [15,45]. In patients with unclear precipitating factors, drug-induced liver injury might be one viable reason, but studies on precipitating factors of ACLF-AKI are lacking [46].
Pathogenesis of acute kidney injury in acute-on-chronic-liver failure
The mechanism of AKI in patients with ACLF is complex and might involve multiple factors such as hemodynamic abnormality, inflammation, and oxidative stress [5,16,47]. Among these, systemic inflammation is a key factor of AKI pathogenesis in ACLF patients [48]. Systemic inflammation in ACLF is usually induced by damage-associated molecular patterns (DAMPs) and body fluids containing pathogen-associated molecular patterns (PAMPs) [49–51]. Bacterial translocation often occurs in cirrhotic patients due to intestinal bacterial overgrowth, increased intestinal mucosa permeability, and impaired intestinal innate immune, and these PAMPs may be a main cause of ACLF [52–54]. However, alcohol-induced DAMPs can be activated in the absence of infection, especially in severe alcoholic hepatitis, and can lead to increased endoplasmic reticulum stress and hepatocyte injury [55,56].
AKI in cirrhosis is initially caused by decreased effective vascular volume due to splanchnic vasodilatation and renal hypoperfusion. However, prolonged duration of AKI is associated with gradual renal parenchymal damage, leading to capillary leukocyte infiltration, vascular microthrombosis, and cell apoptosis [57,58]. In particular, parenchymal damage of the kidney is frequently found in a more advanced stage of AKI or HRS-AKI and is closely related to the low response rate to the treatment with terlipressin plus albumin (i.e., less than 50%) [28,59]. That is, splanchnic vasodilatation and renal artery vasoconstriction may be involved in the pathogenesis of AKI in the early stage, while renal parenchymal damage may also be an important element of AKI pathogenesis in the later stages. Recently, it has been also reported that bile cast nephropathy may play an important role in the pathogenesis of AKI in patients with prolonged jaundice, and further studies are needed [60].
Biomarkers of acute kidney injury in acute-on-chronic-liver failure
The mortality rate is higher in ACLF patients with AKI than in those without AKI [27]. Many studies on biomarkers of AKI have been conducted. However, almost all reported biomarkers for diagnosis and prognosis of AKI were identified in heterogeneous populations comprising various stages of AKI; caution is required in their interpretation. Many studies have been published on biomarkers of AKI in cirrhosis patients, while only three biomarkers of AKI in patients with ACLF have been reported: cystatin C, neutrophil gelatinase-associated lipocalin (NGAL), and serum creatinine. The analysis of 429 patients in the CANONIC study cohort showed that baseline cystatin C was a useful biomarker predicting the development of renal dysfunction and ACLF, which was directly related to mortality [61]. Also, cystatin C was a more effective predictor of renal dysfunction than creatinine in ACLF of HBV patients [62]. However, NGAL did not predict the occurrence of renal dysfunction or ACLF although it was associated with short-term mortality [61]. Interleukin-18 and kidney injury molecule-1 have been examined in cirrhosis patients without ACLF but not in ACLF patients [63]. One of the major limitations of the studies on AKI biomarkers is that cystatin C or NGAL may be affected by the degree of inflammation [24]. In addition, specific cut-off values for the overlapping phenotypes were not available, limiting clinical utility [63].
Last, serum creatinine, which has been traditionally used, can also be an important biomarker in HRS-AKI patients. In HRS-AKI patients with ACLF, a higher level of serum creatinine was associated with lower response rate to terlipressin [64]. Therefore, serum creatinine is an important predictor of response to treatment when HRS-AKI occurs in ACLF patients.
Prevention of acute kidney injury
In cirrhosis patients, AKI prevention plays a critical role in prognosis. Avoidance of nephrotoxic drugs and appropriate control of volume status are crucial. The problem is that it is very difficult to control the volume status in patients with cirrhosis and decreased renal function [65]. NSAIDs and antibiotics are highly likely to promote AKI in cirrhosis patients should be administered with extra caution and careful determination of dose [66]. There is no evidence that contrast-induced nephropathy occurs more frequently in cirrhotic patients than in the general population. Nevertheless, caution is always required because cirrhotic patients require more frequent surveillance or examination than the general population [67]. In addition, frequent follow-up with serum creatinine is necessary in cirrhotic patients with ascites because of the high risk of AKI.
Antibiotic prophylaxis may help prevent AKI. Prophylactic antibiotics are administered in patients with frequent spontaneous bacterial peritonitis (SBP) or variceal bleeding. Systemic inflammation frequently caused by bacterial translocation has been pointed out as the main cause of acute exacerbation; hence, antibiotic prophylaxis that prevents repeated SBP can also prevent AKI [68,69]. Antibiotic prophylaxis is also recommended in patients with varix bleeding to reduce the likelihood of systemic infection and to increase survival [70,71]. In both instances (i.e., SBP and variceal bleeding), there were promising findings that antibiotic prophylaxis could lower the incidence of AKI [72,73].
Albumin infusion may help prevent AKI in two ways. First, large volume paracentesis can induce AKI by causing circulatory dysfunction and hypovolemia; hence, albumin replacement is essential to prevent AKI during large volume paracentesis [74]. Second, albumin is also recommended to prevent AKI in patients with SBP [75]. However, the clinical utility of albumin might be limited in systemic infections other than SBP [8]. A recent study showed that weekly albumin administration can be helpful for long-term prognosis of cirrhosis, demonstrating the anti-inflammatory effect of albumin beyond its volume replacing effect [76]. However, more empirical evidence is needed to safely administer weekly albumin in patients with liver cirrhosis considering cost-effectiveness.
Last, there was a study showing that administration of N-acetylcysteine (NAC) can help prevent AKI in alcoholic hepatitis patients [77]. In addition, one study reported that short-term granulocyte colony-stimulating factor (G-CSF) plus erythropoietin (darbepoetin) can decrease AKI, sepsis, and mortality in DC patients [78]. The effectiveness of NAC or G-CSF has not been clearly demonstrated, and additional results are needed.
Treatment of acute kidney injury in acute-on-chronic-liver failure
First, if the underlying cause of AKI is evident, treatment of the underlying condition needs to be prioritized. For example, antibiotic treatment can be considered for AKI caused by bacterial infection, volume replacement can be administered to treat hypovolemia due to gastrointestinal bleeding, and any causative agent needs to be discontinued to prevent AKI. Also, prednisolone therapy can be considered for severe alcoholic hepatitis [79]. If these underlying conditions are addressed as early as possible, AKI progression may be suppressed. Subsequently, albumin infusion is recommended for volume expansion. Usually, 1 g of albumin per kg of body weight is recommended for 1 to 2 days, after which the improvement of AKI can be evaluated [20]. If there is no response to albumin infusion, additional biomarker testing such as urine NGAL might be considered [80]. If the level of urine NGAL is elevated, it is most likely a phenotype of acute tubular necrosis [63,81].
When AKI continues to progress despite these treatments, vasoconstrictors such as terlipressin with albumin may be considered. However, the use of vasoconstrictor is not recommended for stage I AKI [8]. Treatment response to terlipressin in patients with ACLF accompanying HRS-AKI is reported to be around 35% [19,82]. The response rate to terlipressin is particularly low in patients with HRS-AKI when the baseline serum creatinine is elevated, bilirubin level is heightened, or mean arterial pressure is not elevated despite terlipressin administration [28,83]. If there is no improvement in serum creatinine after 7 to 10 days of terlipressin treatment, it should be discontinued.
ACLF patients with AKI have a high mortality rate; hence, liver transplantation (LT) should be considered. Usually, the prognosis after LT in the presence of one or two organ failures is not different from that of patients without ACLF [84]. However, when there are three or more organ failures, the 1-year posttransplantation survival rate tends to decrease slightly to 80%. Nevertheless, it is recommended to consider LT first in patients with grade 3 ACLF, because the survival rate is less than 20% if LT is not performed [85,86].
Liver-kidney co-transplantation might be a viable option for patients with estimated GFR (eGFR) of <30 mL/min/1.73 m2 or underlying CKD, who are highly likely to need dialysis later [80,87]. However, studies on liver-kidney co-transplantation in ACLF patients are lacking due to donor shortage. In patients who underwent LT only, the rate of renal dysfunction after transplantation was increased proportionally to the pretransplantation AKI duration [88,89]. In particular, patients with AKI for more than 6 weeks were more likely to experience renal dysfunction after LT [90]. Therefore, liver-kidney co-transplantation should be considered first in the earlier-mentioned conditions [16]. In updated Organ Sharing/Organ Procurement and Transplantation Network criteria, liver-kidney co-transplantation is recommended for the following three conditions [91–93]: 1) CKD with a measured or eGFR of <60 mL/min/1.73 m2 for more than 90 consecutive days, 2) sustained AKI, or 3) accompanying metabolic disease such as hyperoxaluria or atypical hemolytic uremic syndrome.
RRT can be performed as a bridging therapy if AKI continues to worsen in patients for whom immediate LT is not available [94,95]. Studies reported that continuous RRT rather than intermittent RRT improves cardiovascular stability and reduces intracranial pressure in patients with cirrhosis [96–98]. However, there is no randomized controlled trial comparing continuous and intermittent RRT, and RRT by itself does not reduce mortality in ACLF or cirrhotic patients.
Last, artificial liver treatment such as that with a Molecular Adsorbent Recirculating System (MARS) can be considered as a bridging therapy in ACLF patients, although it may not improve survival [99,100]. Due to its limited effectiveness and high cost, MARS is not widely used.
Conclusion
ACLF is an acute exacerbation of preexisting liver disease, resulting in high mortality and high socioeconomic burden. In patients with ACLF, the kidney is the most common site of organ failure, and the prognosis is poor when AKI occurs. AKI in patients with ACLF is clinically different from AKI in patients with decompensated liver cirrhosis, but more research is needed to elucidate its mechanism. AKI associated with ACLF develops through multifactorial pathogeneses involving both hemodynamic abnormality and inflammation, especially systemic inflammation, bacterial translocation, and infection. Prophylactic antibiotic administration or albumin replacement might be effective in preventing AKI. Treatment for AKI includes management of underlying disease, volume replacement, vasoconstrictor with albumin, RRT, and LT. More studies are needed to advance the understanding of ACLF-AKI and its prevention and treatment.
Notes
Conflicts of interest
All authors have no conflicts of interest to declare.
Funding
This research was funded by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (2021R1G1A1007886), and in part by the Soonchunhyang University Research Fund.
Data sharing statement
The data presented in this study are available on request from the corresponding author.
Authors’ contributions
Conceptualization, Funding acquisition: SGK
Investigation: JJY
Resources: MYP
Writing–original draft: JJY
Writing–review & editing: SGK
All authors read and approved the final manuscript.