Risk factors for overcorrection of severe hyponatremia: a post hoc analysis of the SALSA trial
Article information
Abstract
Background
Hyponatremia overcorrection can result in irreversible neurologic impairment such as osmotic demyelination syndrome. Few prospective studies have identified patients undergoing hypertonic saline treatment with a high risk of hyponatremia overcorrection.
Methods
We conducted a post hoc analysis of a multicenter, prospective randomized controlled study, the SALSA trial, in 178 patients aged above 18 years with symptomatic hyponatremia (mean age, 73.1 years; mean serum sodium level, 118.2 mEq/L). Overcorrection was defined as an increase in serum sodium levels by >12 or 18 mEq/L within 24 or 48 hours, respectively.
Results
Among the 178 patients, 37 experienced hyponatremia overcorrection (20.8%), which was independently associated with initial serum sodium level (≤110, 110–115, 115–120, and 120–125 mEq/L with 7, 4, 2, and 0 points, respectively), chronic alcoholism (7 points), severe symptoms of hyponatremia (3 points), and initial potassium level (<3.0 mEq/L, 3 points). The NASK (hypoNatremia, Alcoholism, Severe symptoms, and hypoKalemia) score was derived from four risk factors for hyponatremia overcorrection and was significantly associated with overcorrection (odds ratio, 1.41; 95% confidence interval, 1.24–1.61; p < 0.01) with good discrimination (area under the receiver-operating characteristic [AUROC] curve, 0.76; 95% CI, 0.66–0.85; p < 0.01). The AUROC curve of the NASK score was statistically better compared with those of each risk factor.
Conclusion
In treating patients with symptomatic hyponatremia, individuals with high hyponatremia overcorrection risks were predictable using a novel risk score summarizing baseline information.
Introduction
Hyponatremia is the most common electrolyte imbalance encountered in clinical practice, with a prevalence of 14% to 42% in hospitalized patients. Moreover, hyponatremia has a high mortality rate and long hospitalization period [1,2]. Hyponatremia can induce various clinical manifestations that range from mild (fatigue, nausea, vomiting, headache, gait disorder, and confusion) to severe symptoms (seizures, coma, and brain hypoxia) [3–6]. Hypertonic saline has been used to treat symptomatic hyponatremia [7,8]. An increase in serum sodium levels by 4 to 6 mEq/L is generally sufficient to improve the symptoms caused by cerebral edema. However, overcorrection of hyponatremia may result in irreversible neurologic disability such as osmotic demyelination syndrome (ODS) [9–12].
According to an American expert panel recommendation, overcorrection is defined as increase in serum sodium levels of >10–12 mEq/L in any 24 hours period or >18 mEq/L in any 48 hours period, with a more stringent limit of >8 mEq/L in 24 hours for patients at a high risk of developing ODS [13]. A European clinical practice guideline defined overcorrection as an increase in serum sodium levels of >10 mEq/L during the first 24 hours or >8 mEq/L in any 24 hours period thereafter [1]. The distinction between both guidelines indicates that there is still no consensus regarding the definition of hyponatremic overcorrection [1,10,13–17]. Nevertheless, the incidence of overcorrection has been reported to be as high as 20% to 41% [10,14,18–21].
Several studies and guidelines have recommended a specific amount and rate of hypertonic saline for effective hyponatremia treatment and overcorrection prevention [1,10,13,16,18,20,22–24]. However, it remains unclear why overcorrection occurs in some patients that received the required amount of hypertonic saline at the recommended rate. A few studies have examined the risk factors for overcorrection following symptomatic hyponatremia treatment, and these studies were limited by their retrospective design [14,17,18,21]. Therefore, we conducted a post hoc analysis of a prospective randomized controlled study, in which we investigated risk factors for overcorrection in patients receiving treatment for severe hyponatremia. Our goal was to enable physicians to recognize individuals at a high risk of overcorrection and to prevent ODS by careful correction with hypertonic saline.
Methods
Study population
We performed a post hoc analysis of a multicenter, prospective randomized controlled study: the SALSA (Efficacy and Safety of Rapid Intermittent Correction Compared With Slow Continuous Correction With Hypertonic Saline In Patients With Moderately Severe or Severe Symptomatic Hyponatremia) trial. The study included 178 participants who were admitted at Seoul National University Bundang Hospital, SMG-SNU Boramae Medical Center, and Hallym University Dongtan Sacred Heart Hospital between August 2016 and August 2019. The detailed study protocol has been described elsewhere (ClinicalTrials.gov; NCT02887469) [19].
Patients aged above 18 years, with moderate to severe symptoms, and with glucose-corrected serum sodium levels of ≤125 mEq/L were included in this study [25]. Patients with the following conditions were excluded: pseudohyponatremia (serum osmolality, >275 mOsm/kg), primary polydipsia (urine osmolality, ≤100 mOsm/kg), current pregnancy, breastfeeding, anuria, arterial hypotension (systolic blood pressure, <90 mmHg and mean arterial pressure, <70 mmHg), liver disease (transaminase levels of >three times the upper limit of normal, known decompensated liver cirrhosis with ascites or diuretic use, hepatic encephalopathy, and esophageal varices), uncontrolled diabetes mellitus (glycated hemoglobin, >9), a history of cardiac surgery, acute myocardial infarction, sustained ventricular tachycardia, ventricular fibrillation, acute coronary syndrome, cerebral trauma, and increased intracranial pressure. This post hoc analysis was approved by the Institutional Review Boards of three centers: Seoul National University Bundang Hospital (No. B-2101-660-101), SMG-SNU Boramae Medical Center (No. 10-2021-6), and Hallym University Dongtan Sacred Heart Hospital (No. 2020-10-012). Written consent was obtained from all participants or a legal guardian, when applicable. The study complied with principles of the Declaration of Helsinki.
Data collection and definitions
Baseline demographics, alcohol consumption information, and anthropometric measurements were recorded. Chronic alcoholism was defined as the consumption of at least 4 and 3 glasses of alcohol per day in males and females, respectively, regardless of the types of alcohol. We assessed the presence of comorbidities such as hypertension, diabetes mellitus, congestive heart failure, and cancer by screening for the I10–I15; E10–14; I11.0, I13.0, I13.2, I50; and C codes based on the International Classification of Disease, the 10th revision as well as by a self-reported or confirmed history of antihypertensive and antidiabetic drug use. The definition of hyponatremia was based on biochemical severity: ‘severe (profound)’ was defined as serum sodium levels of ≤125 mEq/L [1,13]. Clinical manifestations of hyponatremia were divided into moderate and severe based on the clinical presentation of the patient at initial hyponatremia. Moderate symptoms included nausea, drowsiness, headache, general weakness, and malaise. Severe symptoms included vomiting, stupor, coma (Glasgow Coma Scale score, ≤8), and seizures. We determined the underlying cause of hyponatremia using a structured diagnostic approach based on history, physical examination, and laboratory test findings. Patients were divided into five categories: 1) decreased extracellular fluid (ECF) volume due to renal sodium loss (e.g., diuretics, especially thiazides), 2) decreased ECF volume due to nonrenal sodium loss (e.g., gastrointestinal sodium loss or third spacing), 3) increased ECF volume (e.g., heart failure, liver cirrhosis, and nephrotic syndrome), 4) normal ECF volume with adrenal insufficiency, and 5) normal ECF volume fulfilling essential diagnostic criteria for the syndrome of inappropriate antidiuresis (SIAD) [20,26].
Two infusion methods for hypertonic saline have been described in published protocols; rapid intermittent bolus (RIB) and slow continuous infusion (SCI) [19,20,27]. The initial infusion rate was determined based on hyponatremia symptom severity. The treatment guidelines for the two groups are detailed in Supplementary Methods (available online) and Supplementary Fig. 1 (available online). The treatment goals were to increase serum sodium level by 5–9 mEq/L and achieve symptom relief within the first 24 hours, as well as increase serum sodium level by 10–17 mEq/L or to ≥130 mEq/L and to achieve symptom relief within the first 48 hours. Serum sodium levels were measured every 6 hours for 2 days using indirect ion-selective electrodes at the following three centers with the indicated equipment: Seoul National University Bundang Hospital, AU5800 (Beckman Coulter, Indianapolis, IN, USA) and Dimension Vista 1500 (Siemens Healthineers, Erlangen, Germany); SMG-SNU Boramae Medical Center, Modular DP (Roche Diagnostics, Indianapolis, IN, USA) and Unicel DxC 800 (Beckman Coulter); and Hallym University Dongtan Sacred Heart Hospital, AU5800 (Beckman Coulter).
Study outcomes
The primary outcome was the incidence of hyponatremia overcorrection at any given period, which was defined as an increase in serum sodium levels by >12 mEq/L or >18 mEq/L within the first 24 or 48 hours, respectively. The secondary outcomes represented the time-specific increase in cumulative hyponatremia overcorrection rates specified by time. These were defined as an increase in serum sodium levels by >12 mEq/L within 6, 12, and 24 hours.
Statistical analysis
Baseline characteristics and laboratory data are expressed as mean and standard deviation for continuous variables and frequency and percentage for discrete variables. Differences in continuous variables were analyzed using the Student t test and Mann-Whitney test, and the chi-square and Fisher exact tests were used for discrete variables. Univariable logistic regression was used to analyze each variable to identify significant risk factors for hyponatremia overcorrection. We retained variables with p < 0.05 in the multivariable model using backward selection. Odds ratios (ORs) and 95% confidence intervals (CIs) for hyponatremia overcorrection occurrence were calculated after stepwise adjustment for multiple confounders. We multiplied parameter estimates of discrete variables in the model by a constant to obtain scores. We multiplied coefficients of continuous variables by the constant, which then represented a risk score for each unit increase in an individual continuous variable. The risk score was summarized as the arithmetic sum of the points for each variable. We evaluated model discrimination using the area under the receiver-operating characteristic (AUROC) curve to compare our predictive model to an older overcorrection model (The Severe Hyponatremia Overcorrection Risk [SHOR] score) [17]. The SHOR score has eight risk factors for overcorrection: age, vomiting, somnolence, volume overload, initial serum sodium level, initial serum potassium level, urine osmolality, and chest tumor. We adjusted our prospective data to compare the predictive abilities for overcorrection in the SHOR scoring system and our predictive model. Patients aged under 40 years were included in the 40 to 50 years group, and those with serum sodium levels of >116 mEq/L were included in the serum sodium of 114–116 mEq/L group. We defined somnolence as drowsiness or stupor. We obtained two scores (SHOR1 score: somnolence as stupor, SHOR2 score: somnolence as drowsiness), and each SHOR score was compared with that of our predictive model. Variables with p < 0.05 were considered statistically significant. All analyses and calculations were performed using IBM SPSS version 24.0 (IBM Corp., Armonk, NY, USA), and STATA version 14.0 (StataCorp LP, College Station, TX, USA).
Results
Study population
We enrolled 178 patients who were admitted for symptomatic hyponatremia between August 24, 2016 and August 21, 2019. The mean patients’ age was 73.1 ± 12.2 years and 44.9% of the patients were male. The most common causes of hyponatremia were thiazide diuretic use (n = 53 [29.8%]), and SIAD (n = 52 [29.2%]), with almost the same incidence. These were followed by adrenal insufficiency (n = 44 [24.7%]), decreased ECF volume due to nonrenal sodium loss (n = 39 [21.9%]), and increased ECF volume (n = 19 [10.7%]). Five individuals had a history of chronic alcoholism (n = 5 [2.8%]). Forty-four patients (24.7%) had severe symptoms of hyponatremia. Serum sodium and potassium levels at admission were 118.2 ± 5.0 and 4.0 ± 0.8 mEq/L, respectively.
Incidence and risk factors for hyponatremia overcorrection
Hyponatremia overcorrection occurred in 20.8% (37 of 178) of patients. Patients were classified into overcorrection and no-overcorrection groups, and their baseline characteristics are shown in Table 1. Patients in the overcorrection group were more likely to exhibit chronic alcoholism (8.1% vs. 1.4%), severe symptoms (45.9% vs. 19.1%), and thiazide use (43.2% vs. 26.2%) than those in the no-overcorrection group. The overcorrection group demonstrated lower levels of serum sodium, potassium phosphorus, and osmolarity compared with the no-overcorrection group. The overcorrection group also showed higher serum albumin, aspartate aminotransferase, alanine aminotransferase, and total bilirubin levels than those of the no-overcorrection group. The cumulative amount of hypertonic saline administrated during 48 hours was 554 mL. The cumulative amounts of hypertonic saline infused during the first 1/6 hours did not differ between the groups. Interestingly, the cumulative amounts of hypertonic saline administrated for 24/48 hours were significantly smaller in the overcorrection group than that that in the no-overcorrection group (24 hours, 350 mL vs. 416.3 mL, p = 0.045; 48 hours, 388.5 mL vs. 598.1 mL, p < 0.001). The 48 hours urine output was higher in the overcorrection than that in the no-overcorrection group (5,663 mL vs. 3,401 mL, p < 0.001).
We conducted univariable and multivariable logistic regression analyses separately to identify independent risk factors for hyponatremia overcorrection (Table 2). The univariate analysis showed that chronic alcoholism (OR, 6.13; 95% CI, 0.99–38.16; p = 0.05), severe symptoms of hyponatremia (OR, 3.59; 95% CI, 1.66–7.76; p < 0.01), low serum potassium level (OR, 0.37; 95% CI, 0.21–0.65; p < 0.01), low serum sodium level (OR, 0.84; 95% CI, 0.77–0.90; p < 0.01), high total bilirubin level (OR, 3.06; 95% CI, 1.42–6.58; p < 0.01), high albumin level (OR, 2.38; 95% CI, 1.19–4.76; p = 0.02), and thiazide use (OR, 2.14; 95% CI, 1.01–4.54; p = 0.05) were significant risk factors for overcorrection. Multivariate analysis showed that only chronic alcoholism (OR, 15.27; 95% CI, 1.46–159.28; p = 0.02), severe symptoms of hyponatremia (OR, 2.83; 95% CI, 1.14–7.02; p = 0.03), initial serum potassium level (OR, 0.86; 95% CI, 0.79–0.94; p < 0.01), and initial serum sodium level (OR, 0.34; 95% CI, 0.17–0.67; p < 0.01) were significant risk factors for overcorrection after adjusting for all variables.
Novel risk score for hyponatremia overcorrection (NASK score)
We created a scoring system to facilitate the manual calculation of hyponatremia overcorrection risk, and we used coefficients to change each impact to integer scores for four statistically significant variables. We calculated the scores using risk factors for overcorrection based on multivariable analysis as described in Supplementary Table 1 (available online). The NASK (hypoNatremia, Alcoholism, Severe symptoms, and hypoKalemia) score was calculated as the arithmetic sum of the points for each of these variables. The scores were as follows: chronic alcoholism, 7 points; severe symptoms of hyponatremia, 3 points; low serum potassium level of <3.0 mEq/L, 3 points; and initial sodium level of ≤110, 110–115, 115–120, and 120–125 mEq/L were scored as 7, 4, 2, and 0 points, respectively. NASK score was significantly associated with overcorrection (OR, 1.41; 95% CI, 1.24–1.61; p < 0.01) (Fig. 1). The receiver-operating characteristic curves for each risk factor and NASK score with regard to overcorrection are shown in Fig. 2. NASK score had a good discriminatory ability, with an AUROC curve of 0.757 (95% CI, 0.66–0.85; p < 0.01). The AUROC was 0.752 (95% CI, 0.67–0.84; p = 0.88), 0.691 (95% CI, 0.60–0.79; p = 0.27), 0.634 (95% CI, 0.55–0.72; p < 0.01), and 0.533 (95% CI, 0.49–0.58; p < 0.01) for the baseline serum sodium level, baseline serum potassium level, severity of hyponatremia symptoms, and chronic alcoholism, respectively. The AUROC curve of the NASK score was significantly better than that of each risk factor.
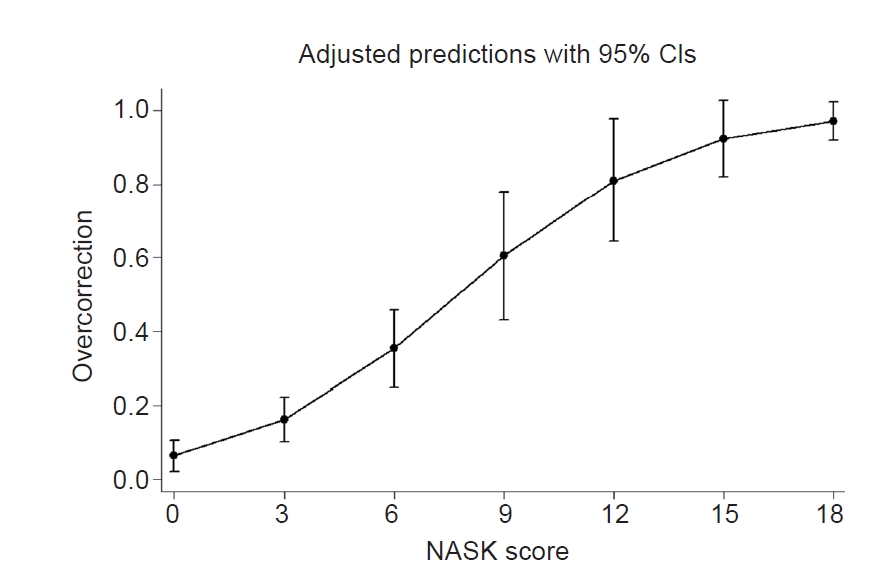
Marginal plot of the NASK score against hyponatremia overcorrection.
CI, confidence interval; NASK, hypoNatremia, Alcoholism, Severe symptoms, and hypoKalemia.
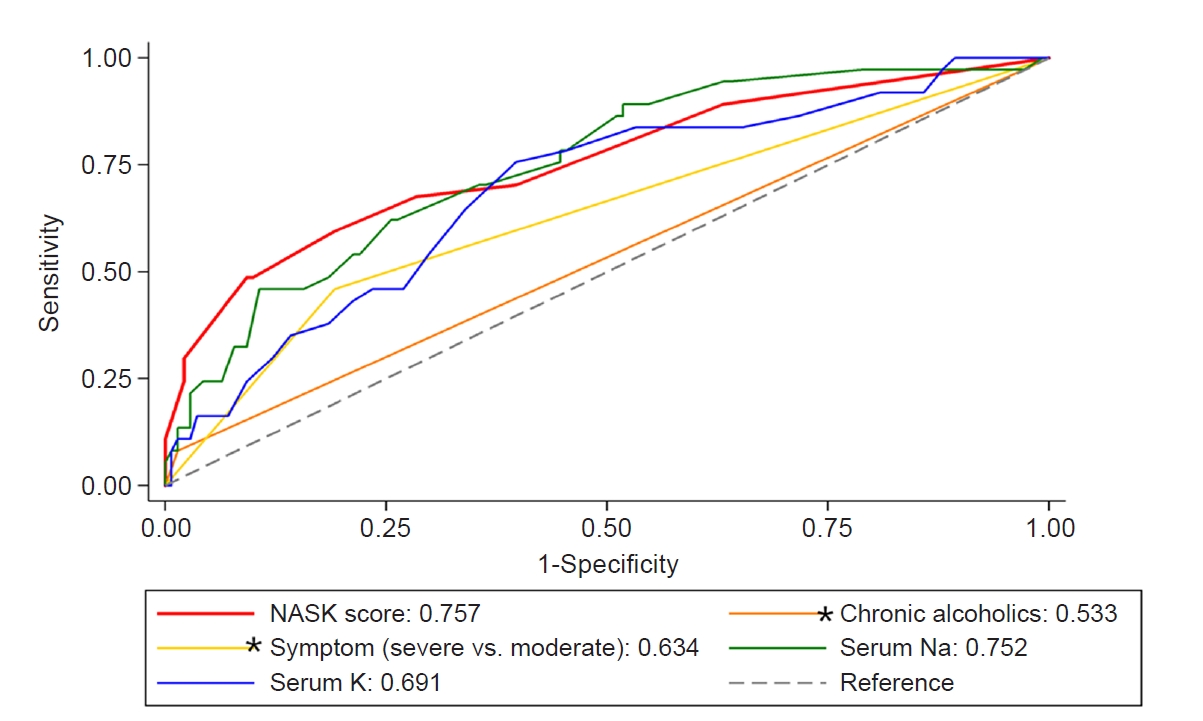
The AUROC curve for the NASK score and each risk factor for hyponatremia overcorrection.
The AUROC curve for each of the two factors (*) is statistically significant when compared to the AUROC of NASK score.
AUROC, the area under the receiver-operating characteristic; NASK, hypoNatremia, Alcoholism, Severe symptoms, and hypoKalemia.
Comparison of the predictive abilities of NASK and SHOR scores
We compared the predictive ability of NASK and SHOR1/SHOR2 scores for hyponatremia overcorrection using AUROC curve analysis (Supplementary Fig. 2, available online). The AUROC curve were 0.757 (95% CI, 0.66–0.85; p < 0.01), 0.722 (95% CI, 0.63–0.82; p = 0.46), 0.722 (95% CI, 0.63–0.82; p = 0.47) for NASK, SHOR1, and SHOR2 scores, respectively. The AUROC curve for NASK score was higher than those of SHOR1 and SHOR2 scores, although there were no significant differences between the values.
Cumulative hyponatremia overcorrection and predictive ability of NASK score by time
We subdivided the overcorrection group by time (within the first 6, 12, 24, and 48 hours) to evaluate whether hyponatremia overcorrection risk factors changed according to time. Overcorrection occurred in 5.1% (n = 9), 6.7% (n = 12), 18.5% (n = 33), and 20.8% (n = 37) of 178 patients within the first 6, 12, 24, and 48 hours, respectively. We analyzed the predictive ability of the four risk factors for overcorrection and NASK score by time based on age, sex, and hypertonic saline infusion method. The four identified risk factors (initial serum sodium level, chronic alcoholism, initial symptoms, and initial serum potassium level) and NASK score had significant predictive abilities for cumulative overcorrection within 6, 12, 24, and 48 hours, excluding initial potassium level within 6 hours and chronic alcoholism within 48 hours (Fig. 3, Fig. 4; Supplementary Table 2, available online).
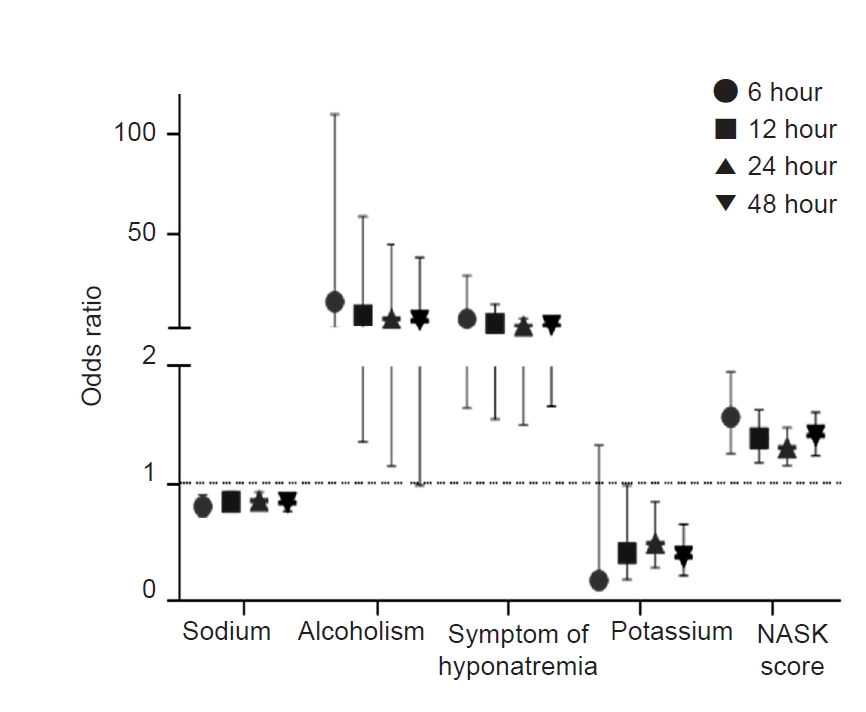
Unadjusted odds ratios of the risk factors for hyponatremia overcorrection.
NASK, hypoNatremia, Alcoholism, Severe symptoms, and hypoKalemia.
Urine output over 48 hours based on hyponatremia overcorrection risk factors
The 48 hours urine output was higher in chronic alcoholics than in non-alcoholics (5,688 mL vs. 3,809 mL, p = 0.08), although the difference was not significant. Furthermore, the urine output was higher in patients with severe symptoms than that of patients with moderate symptoms (5,231 mL vs. 3,419 mL, p < 0.01). Patients with severe hyponatremia had a higher urine output than in those with less severe hyponatremia (6,025, 4,158, 3,767, 3,378 mL for patients with serum sodium levels of ≤110, 110–115, 115–120, and ≥120 mEq/L, respectively; p = 0.02). Moreover, there was a negative correlation between hypokalemia and urine output based on linear regression analysis (β = –0.216, p < 0.01). NASK score also was positively associated with 48 hours urine output (β = 304.5, p < 0.001).
Discussion
In this post hoc analysis of a prospective randomized controlled study, we aimed to evaluate risk factors for hyponatremia overcorrection and to establish a novel scoring system for predicting overcorrection. We identified chronic alcoholism and severe symptoms of hyponatremia as well as lower baseline serum sodium and potassium levels as significant risk factors for hyponatremia overcorrection. The NASK score, an arithmetic sum of the points for each factor after converting the influence of each factor to integer scores, had a higher predictive ability for hyponatremia overcorrection than each factor. Chronic alcoholism, severe symptoms of hyponatremia, low baseline serum sodium levels, and NASK score were risk factors for overcorrection at any time within 48 hours. However, low baseline serum potassium was a risk factor for overcorrection only after 6 hours.
We found that chronic alcoholism, initial serum sodium level, severity of initial symptoms, and initial serum potassium level were significant patient baseline factors that affected the incidence of hyponatremia overcorrection. Alcohol suppresses the endogenous release of antidiuretic hormone (ADH) and occasionally causes free water diuresis when consumed. However, continuous alcohol consumption increases ADH levels, thereby causing water retention. Water and electrolyte retention are resolved within 3 to 6 days after alcohol discontinuation in chronic alcoholics [28,29]. When chronic alcoholics are admitted for hyponatremia, they undergo a period of alcohol withdrawal. Therefore, chronic alcoholics have an increased overcorrection risk during hypertonic saline treatment due to increased diuresis as confirmed by our findings. Previous studies have revealed that lower initial sodium levels [14,17,18,21] and severe symptoms of hyponatremia [17,21] are risk factors for hyponatremia overcorrection, in line with our finding. Hyponatremia overcorrection mainly arises from hypertonic saline treatment or water diuresis. Hyponatremia causes a hypo-osmolar state, leading to a decreased release of ADH and an increase in free water excretion. Supplying hypertonic saline in patients with hyponatremia induces an increase in ADH level and a decrease in water clearance, which occur at a slower rate in patients with severe hyponatremia [30]. However, we cannot explain the mechanism underlying free water excretion using our data because we did not collect urine sodium and potassium levels, as well as osmolality during the 48-hour follow-up. Symptoms of hyponatremia were classified into two groups, and the initial hypertonic saline infusion rate was decided based on symptom severity. A higher amount of hypertonic saline was administered in patients with severe symptoms compared with that in patients with moderate symptoms (317 mL vs. 217 mL, p < 0.01; within the first 6 hours). In line with previous studies [14,17], we found that lower initial potassium levels were associated with hyponatremia overcorrection occurrence. In the Adrogue-Madias formula, replacing potassium plays a significant role in correcting hyponatremia [7,31]. The loss of sodium or potassium induces an osmolar shift to maintain the osmolar balance between the extracellular and intracellular spaces [32]. Potassium loss shifts sodium intracellularly, induces hyponatremia, and enhances ADH release, thereby worsening hyponatremia [33]. Moreover, potassium restriction reportedly increases free water clearance [34]. Therefore, hypokalemia can increase the incidence of hyponatremia overcorrection by increasing diuresis, as confirmed by our findings. Not only each factor but also the NASK score, which combines the scores of these factors, had a positive relationship with the 48 hours urine output.
In addition to the four risk factors identified in our study, previous studies have identified younger age, higher infusion volume, lower urine osmolality, and lower urine sodium levels as risk factors for hyponatremia overcorrection [14,21]. Volume overload and the presence of chest tumor are negatively associated with hyponatremia overcorrection [17]. Woodfine et al. [17] established a novel scoring system (the SHOR score) to predict hyponatremia overcorrection, with risk factors such as age, vomiting, somnolence, volume overload, initial serum sodium level, initial serum potassium level, urine osmolality, and the presence of chest tumor. Our study found the following risk factors for hyponatremia overcorrection: chronic alcoholism, initial serum sodium level, severity of initial symptoms, and initial serum potassium level. Moreover, we developed a scoring system (NASK score) to aid clinicians in quantitatively stratifying an individual’s risk for hyponatremia overcorrection. We compared the predictive ability of hyponatremia overcorrection between the NASK scoring system and the SHOR scoring system [17]. The AUROC curve for NASK score was greater than those for SHOR1 and SHOR2 scores but without statistical significance (Supplementary Fig. 2). Nevertheless, NASK score is easy to calculate as it requires only four factors, whereas SHOR scores require eight discrete factors. Furthermore, NASK score seems to predict hyponatremia overcorrection better than SHOR scores.
This study has several strengths. First, we obtained complete baseline characteristics as well as laboratory data on all patients because of the prospective nature of the original study. We also assessed the cumulative hyponatremia overcorrection rate and verified the predictive ability of each risk factor and the NASK score for the overcorrection rate. Second, in comparison with other retrospective studies, we set up the treatment protocol with hypertonic saline and serum sodium relowering treatment following international guideline recommendations (RIB and SCI). This allowed us to correct the impact of the infusion method of hypertonic saline on treatment outcomes. Therefore, we were able to establish a more explainable scoring system using baseline characteristics. There are several studies or guidelines regarding the adequate amount of hypertonic saline and the required rate for effective hyponatremia treatment and hyponatremia overcorrection prevention. However, inadvertent hyponatremia overcorrection occurs because of unanticipated water diuresis, even when the recommended quantity of hypertonic saline is administrated at a recommended rate or when hypertonic saline administration is stopped. Using this scoring system, physicians can predict hyponatremia overcorrection occurrence before hypertonic saline administration; patients with high NASK scores and increased diuresis can undergo careful monitoring of their status and laboratory data during treatment.
This study also has several limitations. First, due to the prospective design, our study population was smaller than those of other retrospective studies. Second, serum sodium levels were obtained from the three study sites using different measuring machines that were not calibrated. Variabilities in serum sodium assays using each device were inevitable due to the post hoc study design. Third, low sensitivity and positive predictive value were imperative, as the prevalence of chronic alcoholism was low. We additionally performed sensitivity analyses with the NSK (Na, symptoms of hyponatremia, and K) score (total, ≤13) and NK (Na and K) score (total, ≤10) for hyponatremia overcorrection models. The AUROC curves for NSK and NK scores were 0.745 (95% CI, 0.65–0.84; p < 0.01) and 0.721 (95% CI, 0.63–0.81; p < 0.01), respectively. Fourth, the findings were not validated in an external cohort of patients with hyponatremia. A validation study with a larger population should be conducted to confirm our study findings. Specifically, further studies should clarify whether treatment policies should be modified based on the finding that the NASK score influences hyponatremia overcorrection incidence and prognosis. Fifth, some studies have reported a relationship between the occurrences of hyponatremia overcorrection and ODS, which is a critical outcome of hyponatremia overcorrection [11,14,17,24,35,36]. However, there was no ODS in 20.8% of patients with hyponatremia overcorrection, although risk factors for overcorrection were identified to prevent ODS in patients with hyponatremia by reducing overcorrection occurrence. Hyponatremia overcorrection is considered a good laboratory outcome that predicts ODS occurrence because it can be monitored. Moreover, it is a correctable factor during hypertonic saline treatment.
In conclusion, hyponatremia overcorrection occurred in 20.8% of patients who underwent hypertonic saline treatment. The risk factors for hyponatremia overcorrection included chronic alcoholism and severe symptoms of hyponatremia as well as lower initial serum sodium and potassium levels. In patients undergoing symptomatic hyponatremia treatment, overcorrection might be quantitatively predicted using a novel risk score that is summarized by patient baseline information. External validation studies of the NASK score are required to clarify our results.
Supplementary Materials
Notes
Conflicts of interest
All authors have no conflicts of interest to declare.
Funding
The study was supported by grants from the National Research Foundation of Korea (No. 2021R1C1C1008966 and 2019R1A2C1085411).
Authors’ contributions
Conceptualization: HY, SHB, SK
Data curation: SHB, SK
Formal analysis: HY, SHB, SK
Funding acquisition: SHB, SK
Technical and material support: All authors
Visualization: HY, SHB, SK
Writing–original draft: HY, SHB, SK
Writing–review & editing: All authors
All authors read and approved the final manuscript.



