Introduction
Chronic kidney disease (CKD) is a global health problem affecting 8% to 16% of adults [
1]. Defined as decreased kidney function for > 3 months, individuals with CKD are at increased risk of morbidity and early mortality [
1]. Cardiorespiratory fitness is defined as the integrated ability of the circulatory and respiratory systems to supply oxygen during sustained physical activity [
2], and it is widely considered to be the best reflection of whole-body health and function [
3]. Low cardiorespiratory fitness is strongly associated with early all-cause and cardiovascular-related mortality [
3,
4] and presents a higher risk of early death than other common risk factors such as diabetes or hypertension [
5]. Compared to the general population, CKD patients demonstrate a large reduction in cardiorespiratory fitness that occurs early in the disease process and declines concomitantly with renal function [
6,
-
8]. In patients with CKD, low cardiorespiratory fitness is associated with increased aortic stiffness and left ventricle afterload, poor left ventricle function, and a higher burden of cardiovascular risk [
8], and it is unsurprisingly an independent predictor of early mortality [
9,
10]. Many patients with renal disease have cardiorespiratory values comparable to those with mild heart failure [
11].
Cardiopulmonary exercise testing (CPET) is a universal assessment of cardiorespiratory fitness that uses a breath-by-breath gas exchange analysis to provide an objective and accurate measurement of oxygen uptake [
3]. Although the measurement of maximal oxygen uptake (⩒O
2max) is the most reliable measure of exercise capacity, this value is seldom achieved because it demands maximal effort from the individual being tested and a plateau in oxygen uptake despite an increasing exercise workload, which limits its usefulness in groups such as the elderly and those with chronic disease [
12]. Consequently, in such groups and in patients with renal disease, cardiorespiratory fitness is typically derived from ⩒O
2peak because many patients prematurely stop the test due to fatigue or breathlessness [
6]. ⩒O
2peak is also limited by the strong influence of motivation and the subjective choice of a test endpoint [
12].
Baba et al [
13] developed an objective, reproducible estimation of cardiorespiratory function called the oxygen uptake efficiency slope (OUES) that does not require a maximal exercise effort and might therefore be useful in populations, such as CKD patients, deemed high risk for maximal exercise testing [
7]. The OUES integrates cardiovascular, musculoskeletal, and respiratory function into a single index [
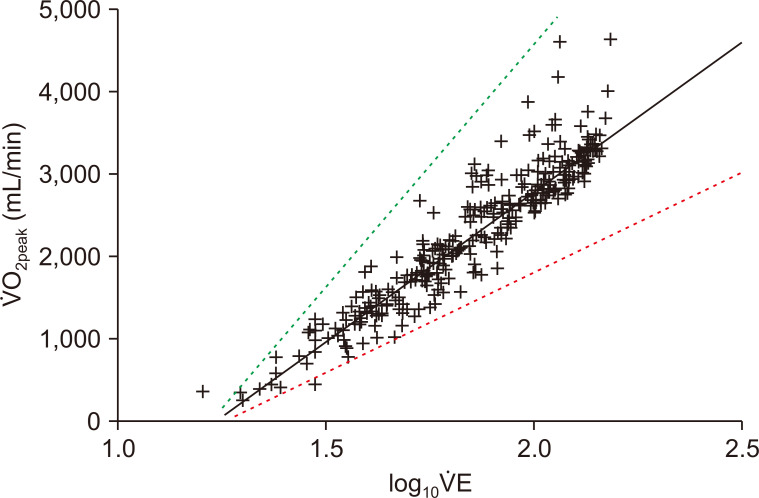
14]. Remaining relatively stable across CPET duration, the OUES represents the relationship (and efficiency) between oxygen uptake and total ventilation and provides an indication of cardiorespiratory reserve, so it could be a more pragmatic and safer alternative to maximal exercise testing in high-risk patients [
7]. In patients with heart failure, the OUES has superior independent prognostic value over other commonly reported CPET-derived values such as ⩒O
2peak and the ⩒E/⩒CO
2 slope [
15]. Although research on the subject is scarce, Kirkman et al [
7] showed a 16% reduction in the OUES in CKD patients compared to healthy controls.
Generally, although not always [
16,
17], exercise training can increase ⩒O
2peak in patients with CKD [
17,
18]. However, little is known about the effect of exercise interventions on the OUES in any population. In patients with chronic heart failure (CHF) [
14,
19] or coronary artery disease (CAD) [
20,
21] and those undergoing hemodialysis (HD) [
22], the OUES increased by 17% to 21% following exercise. An increase in the OUES suggests that a given oxygen uptake is achieved with lower ventilatory cost [
21], although not all research supports those findings [
23].
In this secondary analysis of previous research [
16], we explore the response of the OUES to a 12-week exercise training program in patients with CKD. We hypothesized that 12 weeks of exercise would increase the OUES without necessarily changing the ⩒O
2peak, indicating improvements in oxygen utilization efficiency.
Discussion
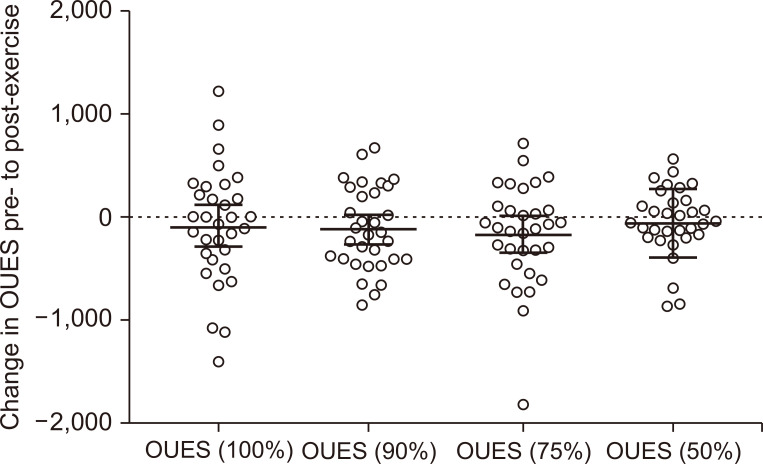
This is the first study to describe the effects of exercise on the OUES in patients with CKD. Our results show that 12 weeks of exercise had no beneficial effects on the OUES, and no meaningful differences appeared between the exercise modalities tested. The lack of change in the OUES supports the insignificant and modest change observed in ⩒O2peak. Exercise-induced responses in the OUES appeared to be more variable than changes in traditional parameters such as ⩒O2peak. The lack of change could indicate a dysfunctional cardiorespiratory response to exercise in patients with CKD.
Since the introduction of the OUES [
13], its use as an objective, reproducible estimation of cardiorespiratory function in clinical populations has grown. Integrating cardiovascular, musculoskeletal, and respiratory function into a single index [
14], the OUES represents the relationship between oxygen uptake and total ventilation. Given its linear presentation, the advantage of the OUES is its ability to obtain a valid measure with submaximal effort, which reduces reliance on parameters such as ⩒O
2peak that are strongly influenced by motivation and the choice of test endpoint [
12]. Research exploring the role of the OUES in kidney disease has been limited. Kirkman et al [
7] found that the OUES of 31 patients was 16% lower than that of healthy controls. Suggestive of further underlying cardiovascular dysfunction, other parameters, such as ⩒O
2peak, VT, ⩒E/⩒CO
2, and ⩒E/⩒O
2, were also impaired. Similarly, research by Tsuyuki et al [
24] found a reduced OUES in HD patients. Given its prognostic value [
15] and use as a potential marker of cardiorespiratory function, efforts to improve OUES values could be beneficial.
Given the lack of major differences between exercise modality groups, we focus on all patients as one cohort in this discussion. We observed a modest, nonsignificant increase in ⩒O
2peak of 0.8 mL/min kg
-1 or ~4%. This result is well below the generally accepted minimal clinically important difference of 1.5 mL/min kg
-1 [
25] and supports the conclusions of some previous exercise studies in CKD that showed inadequate change [
26,
27]. Nevertheless, it does contrast with most of the findings from a recent systematic review and meta-analysis that reported a pooled increase of 2.4 (1.0 to 3.8) mL/min kg
-1 (~11%) in patients with CKD following exercise [
17]. Notably, we found that the increase after exercise was identical to that observed during the control period. Previous work by our group has shown that the minimal detectable change in ⩒O
2peak is 0.5 mL/min kg
-1 [
25], and as such, the change in ⩒O
2peak observed here is likely due to methodological limitations and the error inherent in this test. Improvements in relative ⩒O
2peak (i.e., adjusted for kg) could also be explained by the concurrent changes in body mass seen throughout the study. That possibility is supported by the complete lack of change when ⩒O
2peak is expressed in liters.
We expected the OUES to overcome the limitations associated with ⩒O
2peak measurement. However, contrary to our original hypothesis, we observed no changes in the OUES calculated at any stage of test duration in either group. This includes OUES (75%), which was proposed [
13] to replace ⩒O
2peak or OUES (100%) because it does not rely on maximal effort. Our observations extend the results of Mourot et al [
23], who reported that, despite an increase in ⩒O
2peak, no change occurred in the OUES of healthy young women following 6 weeks of exercise. However, our results contrast with previous research in CHF [
14,
19], CAD [
20,
21], and HD [
22] that has shown improvements in the OUES in the region of 17% to 21%, with changes in the OUES corresponding to improvements in ⩒O
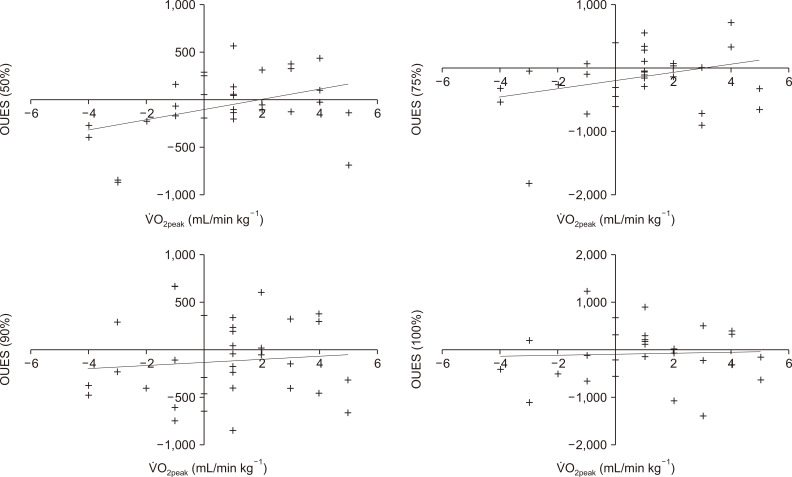
2peak. Interestingly, changes in the OUES corresponded to changes in oxygen uptake but only during the first half of the exercise test. At the end of the test, changes in the OUES and ⩒O
2peak had no relationship. This finding could be due to the submaximal intensity of the first part of the test or variable attainments of true peak values in this group.
Many mechanisms can influence the OUES, including physiologic pulmonary dead space, which depends on the structural integrity of the lungs and the adequacy of pulmonary perfusion; the PaCO2 set-point; and the timing and degree of metabolic acidosis, namely, the amount of blood perfusion to the working muscles [
12,
19,
22]. Consequently, a large (superior) OUES depends on a substantial mass of working muscle, a vigorous and unimpaired blood flow to those muscles, efficient extraction and use of oxygen by those muscles, and the delayed appearance of lactic acidosis [
12]. As such, improvements in the OUES in response to exercise are thought to be driven primarily by peripheral metabolic adaptations, such as increased aerobic metabolism [
20], specifically increases in capillary density, muscle blood flow, and mitochondrial density that delay the onset of metabolic acidosis, thereby decreasing the ventilatory response during exercise [
19,
-
21].
The absence of change in the OUES of our CKD patients could be caused by a myriad of factors, although is likely due to dysfunctional peripheral metabolic mechanisms. Skeletal muscle mitochondrial abnormalities have been noted in patients with CKD [
28]. To date, data from human studies are lacking, but skeletal muscle biopsies taken from CKD patients have revealed reduced mitochondrial density and mitochondrial DNA copy numbers, abnormal morphology, and reduced activity of pyruvate dehydrogenase [
29], a key coupling enzyme in cellular respiration [
30]. Dysfunctional mitochondria are an important source of reactive oxygen species, implicated in cardiovascular morbidity and mortality [
31,
32]. Patients with mitochondrial dysfunction are unable to adequately use O2 for oxidative phosphorylation; instead, lactic acid accumulates early in exercise, which leads to exaggerated circulatory and ventilatory responses [
33]. Research by our group showed greater than normal deoxygenation of skeletal muscle hemoglobin in CKD patients during an incremental exercise test, which we hypothesized to indicate reduced mitochondrial and oxidative phosphorylation capacity [
34]. We have also recently shown that at the cellular level, exercise training has no effect on mitochondrial mass or the mRNA expression of key proteins involved in mitochondrial biogenesis [
35]. This points toward an intrinsic defect in our population in regard to their mitochondrial function.
Metabolic acidosis is a common complication in patients with CKD [
36], and it has been implicated in the induction of mitochondrial dysfunction [
36]. Early onset of lactate production during exercise testing has previously been observed in HD patients [
37], and although research in earlier stages of CKD is limited, Sprick et al [
38] found that metabolic acidosis contributes to exercise intolerance in CKD by promoting earlier fatigue and exacerbation of blood pressure reactivity during exercise. In deconditioned patients (such as those with chronic disease) who develop lactic acidosis early during exercise, the OUES will be expected to be diminished [
12]. Exercise training has been shown to inhibit exercise-induced metabolic acidosis in healthy individuals and in those with CHF [
22], but in the uremic milieu associated with CKD, this response could be diminished.
In our study, CKD-related peripheral limitations on exercise capacity are further indicated by an elevated ventilatory cost of oxygen uptake (⩒E/⩒O
2) compared with values found in those without CKD [
7]. In individuals with mitochondrial myopathies, steep ⩒E/VO2 slopes are observed because O2 delivery exceeds the capacity for utilization [
38]. The ability of peripheral vessels to dilate in response to increased shear stress during exercise has emerged as an important determinant of exercise capacity. In patients with CKD, vascular endothelial dysfunction, as evidenced by increased arterial stiffness, is thought to contribute to exercise intolerance [
39,
40]. The lack of change in the OUES could also be caused by the highly variable response of this value to exercise, and it might not be a sensitive marker in CKD. This supports conclusions already drawn by others [
23].
We observed little response to exercise in the other cardiorespiratory parameters we tested, although we did observe an increase in ⩒E and RR. The increase in ⩒E was found in the AE group only, and increased RR was identified in both groups. ⩒E is a product of both tidal volume and RR [
33]; therefore the increase seen in ⩒E following exercise is likely partly explained by the increase in RR during the test. Following the intervention, the duration of the CPET increased, which might have increased the effort required to sustain the desired workload. This increased duration might also have required increased ⩒E and RR. An increased peak ⩒E (and/or RR) value with a relatively invariable VO2, as observed in our sample, could also be characteristic of inefficient breathing patterns [
33]. Despite an increase in VE, we observed no change in the ⩒E/⩒CO
2 slope, suggesting no improvement in CO2 exhalation and no reduction in lactic acid production [
19]. Kirkman et al [
7] previously showed an increased ⩒E/⩒CO
2 slope in patients with CKD versus healthy controls. The high ⩒E/⩒CO
2 slope observed in our sample could represent impaired ventilatory efficiency, and our mean values at each timepoint, all > 34, indicate an abnormal ventilatory response [
33,
41] similar to those seen in patients with pulmonary disease and CHF [
33].
Although it is limited by the absence of a non-exercising control group, our study benefits from having a control period in which we could assess normal variations in the parameters reported. This was important because it allowed us to observe increases in ⩒O
2peak comparable to those from the 12-week exercise program, supporting the finding that aerobic capacity was unchanged by exercise. It is unlikely that this was caused by an inadequate training stimulus (i.e., duration or intensity) because significant improvements in body composition, strength, and other physical functions were observed as a result of the program [
16]. We were able to show good reproducibility of the OUES before and after this control period, with the OUES (100%) differing by just 0.4%. As a secondary analysis, the results here might not be powered to detect statistically significant changes, and therefore they need to be confirmed in larger trials with this population.
In conclusion, we were unable to obtain an improvement in the OUES after 12 weeks of exercise in patients with CKD. The OUES was hypothesized to be a useful index that circumvents the methodological limitations of maximal exercise testing in evaluating cardiorespiratory reserve in patients. However, although the heterogeneous variation in the OUES response could partly explain our findings, the lack of change in the OUES supports the limited change in ⩒O2peak and other cardiorespiratory parameters measured. Thus, our findings could indicate a dysfunctional cardiorespiratory response to exercise in patients with CKD.





 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print















