| Kidney Res Clin Pract > Volume 43(2); 2024 > Article |
|
Abstract
Background
Methods
Results
Notes
Funding
This study was supported from the Shin Kong Wu Ho-Su Memorial Hospital Research Foundation (grant no. 2018SKHADR007).
Supplementary Materials
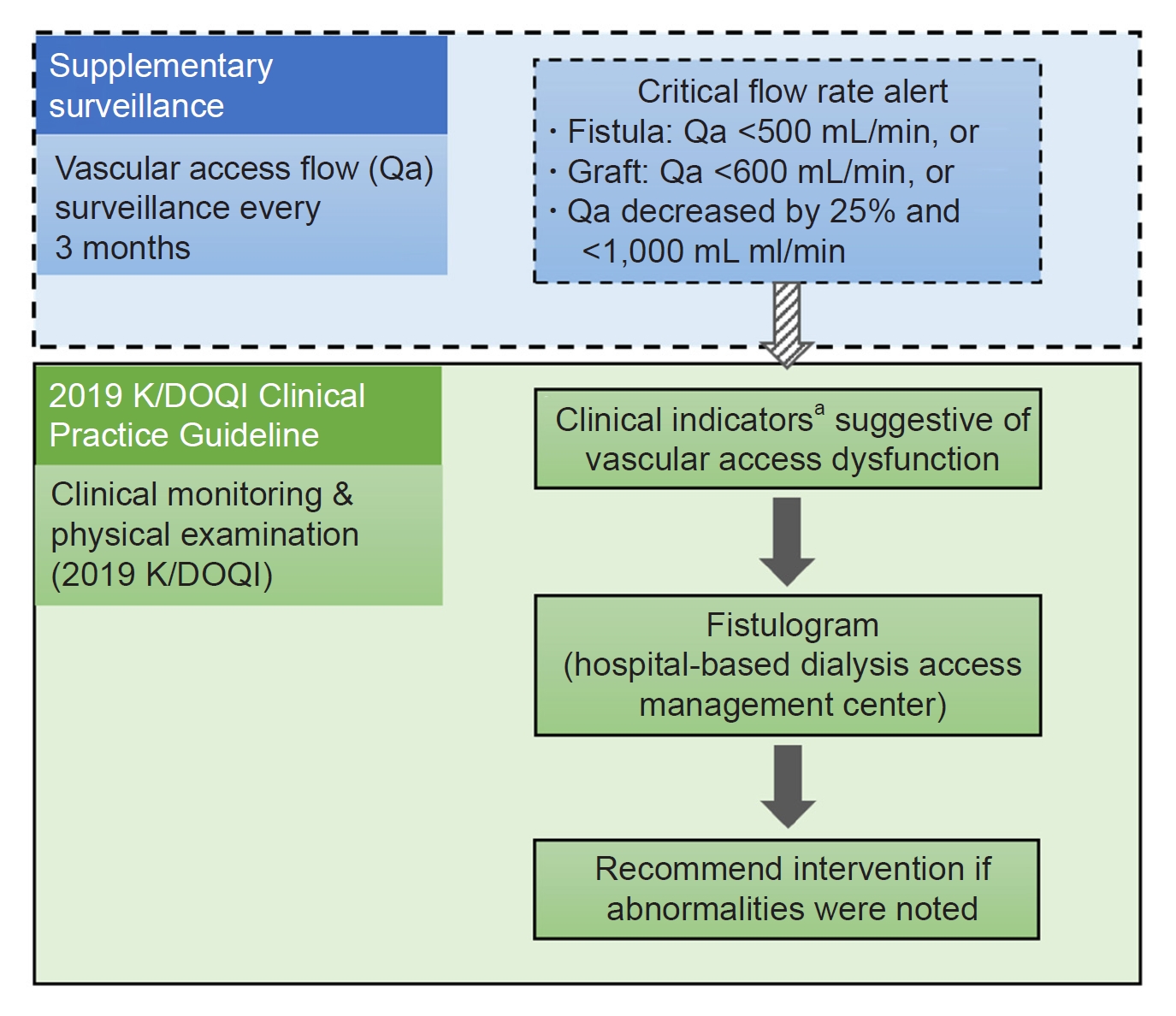
Figure┬Ā1.
Recommended management strategies of the collaborative program involving supplementary routine surveillance.
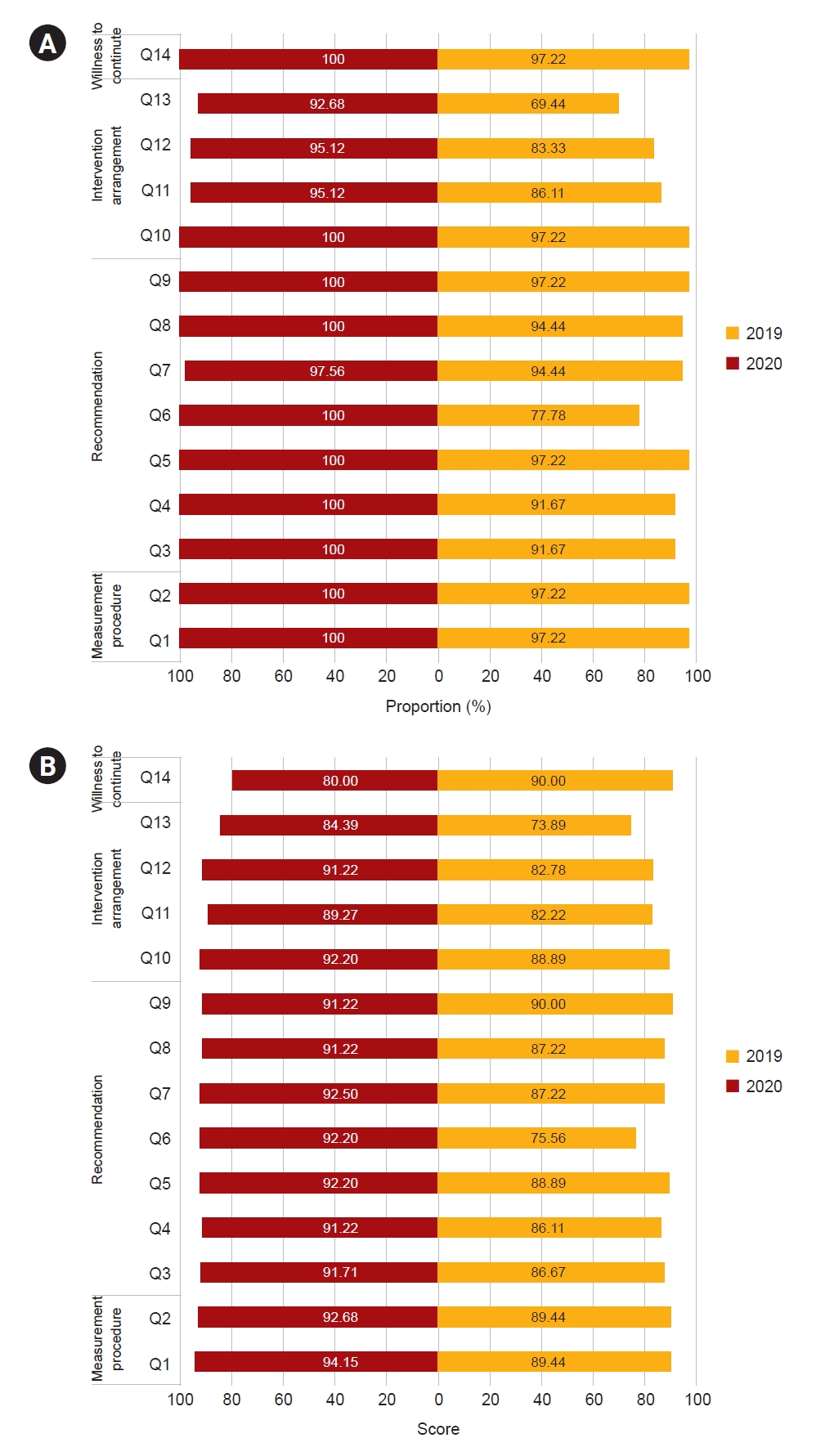
Figure┬Ā2.
Comparison of satisfaction survey responses from clinics between 2019 and 2020.
Figure┬Ā3.
Comparison of the referral rate and distance between hospitals and clinics between 2019 and 2020.

Table┬Ā1.
| Characteristic | AVF | AVG | p-value |
|---|---|---|---|
| No. of subjects | 339 | 53 | |
| Age (yr) | 65.0 (59.0ŌĆō73.0) | 67.0 (61.0ŌĆō77.0) | 0.20a |
| SBP (mmHg) | 140 (123ŌĆō155) | 131 (118ŌĆō146) | 0.03a,* |
| DBP (mmHg) | 70 (61ŌĆō80) | 65 (59ŌĆō75) | 0.03a,* |
| Sex | |||
| ŌĆāFemale | 158 (46.6) | 38 (71.7) | 0.001b,* |
| ŌĆāMale | 181 (53.4) | 15 (28.3) |
Table┬Ā2.
| Variable |
AVF (N = 339) |
AVG (N = 53) |
||||
|---|---|---|---|---|---|---|
| 2019 | 2020 | p-value | 2019 | 2020 | p-value | |
| Ratio of critical flowa | ||||||
| ŌĆāOverall | 207/1,133 (18.3) | 123/972 (12.7) | 0.0004b,* | 44/168 (26.2) | 26/124 (21.0) | 0.30 |
| ŌĆāŌĆāClinic A | 72/460 (15.7) | 47/414 (11.4) | 0.06 | 17/68 (25.0) | 12/56 (21.4) | 0.64 |
| ŌĆāŌĆāClinic B | 23/129 (17.8) | 19/118 (16.1) | 0.72 | 3/17 (17.7) | 4/10 (40.0) | 0.20 |
| ŌĆāŌĆāClinic C | 60/277 (21.7) | 29/238 (12.2) | 0.005b,* | 14/36 (38.9) | 4/29 (13.8) | 0.02b,* |
| ŌĆāŌĆāClinic D | 18/116 (15.5) | 10/101 (9.9) | 0.22 | 1/8 (12.5) | 0/8 (0) | 0.30 |
| ŌĆāŌĆāClinic E | 34/151 (22.5) | 18/101 (17.8) | 0.37 | 9/39 (23.1) | 6/21 (28.6) | 0.64 |
| Vascular access flow | ||||||
| ŌĆāOverall | 1,035.80 ┬▒ 496.76 (n = 1,133) | 1,076.30 ┬▒ 513.65 (n = 972) | 0.07 | 972.38 ┬▒ 416.37 (n = 168) | 1,013.06 ┬▒ 391.44 (n = 124) | 0.40 |
| ŌĆāŌĆāClinic A | 1,101.43 ┬▒ 484.50 (n = 460) | 1,141.62 ┬▒ 545.87 (n = 414) | 0.25 | 998.53 ┬▒ 426.09 (n = 68) | 1,074.82 ┬▒ 445.51 (n = 56) | 0.33 |
| ŌĆāŌĆāClinic B | 1,063.64 ┬▒ 441.91 (n = 129) | 1,100.17 ┬▒ 507.73 (n = 118) | 0.55 | 956.47 ┬▒ 182.82 (n = 17) | 769.00 ┬▒ 223.68 (n = 10) | 0.03c,* |
| ŌĆāŌĆāClinic C | 972.38 ┬▒ 474.54 (n = 277) | 991.89 ┬▒ 423.28 (n = 238) | 0.63 | 778.06 ┬▒ 369.95 (n = 36) | 884.48 ┬▒ 229.71 (n = 29) | 0.16 |
| ŌĆāŌĆāClinic D | 1,172.76 ┬▒ 636.84 (n = 116) | 1,159.50 ┬▒ 579.97 (n = 101) | 0.87 | 1,413.75 ┬▒ 478.51 (n = 8) | 1,366.25 ┬▒ 390.93 (n = 8) | 0.83 |
| ŌĆāŌĆāClinic E | 823.18 ┬▒ 413.19 (n = 151) | 896.34 ┬▒ 440.00 (n = 101) | 0.18 | 1,022.56 ┬▒ 422.81 (n = 39) | 1,007.62 ┬▒ 363.70 (n = 21) | 0.89 |
| Clinical event ratio | ||||||
| ŌĆāNo. of PTAs | 0.324 (n = 110) | 0.316 (n = 107) | 0.80 | 0.774 (n = 41) | 0.509 (n = 27) | 0.005b,* |
| ŌĆāNo. of new AVF creations | 0.003 (n = 1) | 0 (n = 0) | 0.32 | 0 (n = 0) | 0.038 (n = 2) | 0.15 |
| ŌĆāNo. of new AVG creations | 0.006 (n = 2) | 0 (n = 0) | 0.16 | 0.057 (n = 3) | 0 (n = 0) | 0.08 |
Data are expressed as number (%), mean ┬▒ standard deviation, or ratio only. Event ratio = number of critical flow events / number of records in 1 year.
AVF, arteriovenous fistula; AVG, arteriovenous graft; N, number of participants; n, number of records; PTA, percutaneous transluminal angioplasty.
References
- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 1,564 View
- 85 Download
- ORCID iDs
-
Chung-Kuan Wu

https://orcid.org/0000-0003-4446-0167Yu-Wei Fang

https://orcid.org/0000-0002-1302-1885Chia-Hsun Lin

https://orcid.org/0000-0003-1041-0516 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement table 1
Supplement table 1 Print
Print















