Immune checkpoint inhibitor-induced acute tubulointerstitial nephritis
Article information
A 69-year-old Chinese man was hospitalized for Common Terminology Criteria for Adverse Events (CTCAE) grade 2 acute kidney injury 8 months following treatment initiation with combination cytotoxic T-lymphocyte-associated protein 4 and programmed cell death protein-1 (PD-1) immune checkpoint blockade for treatment of adenocarcinoma of unknown primary. His medical history included immune checkpoint inhibitor (ICI)-induced adenohypophysitis and gastrointestinal reflux disease, for which proton pump inhibitor (PPI) omeprazole 20 mg twice daily was commenced 6 months prior. His serum creatinine (sCr) peaked at 299 μmol/L from a baseline of 100 μmol/L. Urinalysis was notable for pyuria (13 white blood cells/µL) without hematuria or significant proteinuria (urine protein-creatinine ratio, 0.27 g/g). C-reactive protein was elevated at 51.8 mg/L (reference range, 0.2–9.1 mg/L). Serum complements were normal. Virology and autoimmune serologies such as hepatitis B, C, anti-double-stranded DNA, and antineutrophil cytoplasmic antibodies were negative. Ultrasonography revealed normal-sized kidneys without obstruction.
A kidney biopsy showed substantial interstitial inflammation and acute tubular injury consistent with ICI-induced acute tubulointerstitial nephritis (ICI-ATIN) (Fig. 1A, B). The diffuse tubulointerstitial mononuclear inflammatory infiltrates comprised lymphocytes, histiocytes, and some plasma cells, without tissue eosinophilia. No interstitial or intratubular granulomas were present. The glomeruli were relatively uninvolved. Interestingly, electron microscopy demonstrated moderate effacement of the foot processes suggestive of podocyte injury (Fig. 1C).
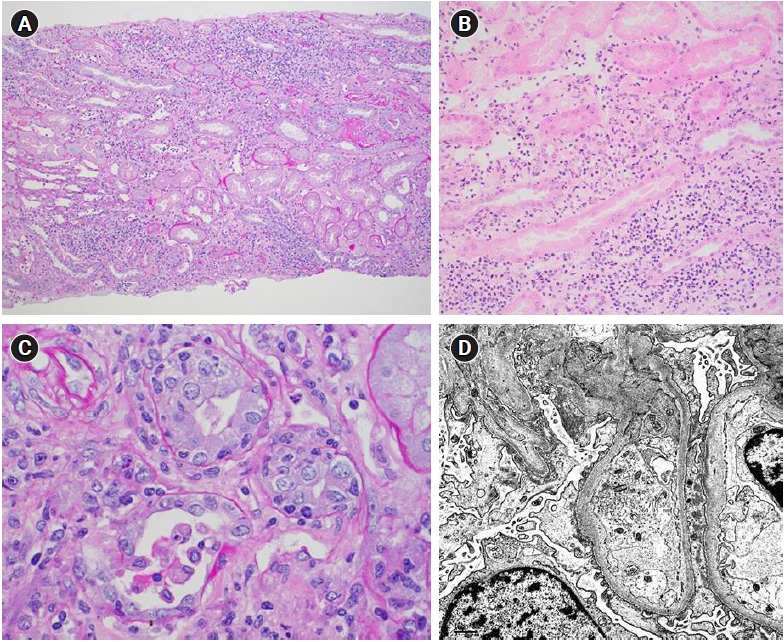
Kidney biopsy showed acute tubulointerstitial nephritis.
(A, B) A diffuse tubulointerstitial mononuclear inflammatory infiltrate was noted, comprising lymphocytes, histiocytes, and some plasma cells, without tissue eosinophilia. No well-formed interstitial or intratubular granulomas are present (periodic acid-Schiff, ×100). (C) Lymphocytic tubulitis (periodic acid-Schiff, ×100). (D) Electron microscopy revealed a moderate degree of podocyte foot process effacement, affecting 30% to 40% of peripheral capillary wall surface area. This suggests an element of podocyte injury. No electron dense deposits were seen (×6,000).
Immunohistochemistry stains showed a predominantly CD8+ lymphocytic infiltrate, macrophages, and scattered plasma cells (Fig. 2). CD3+ T cells constituted about 80% of the total number of lymphocytes, while CD20+ B cells constituted the remaining 20%. About two-thirds of the T cells were CD8+, while one-third were CD4+. CD68+ histiocytes were fairly abundant. CD138+ plasma cells were the least abundant inflammatory cell type.
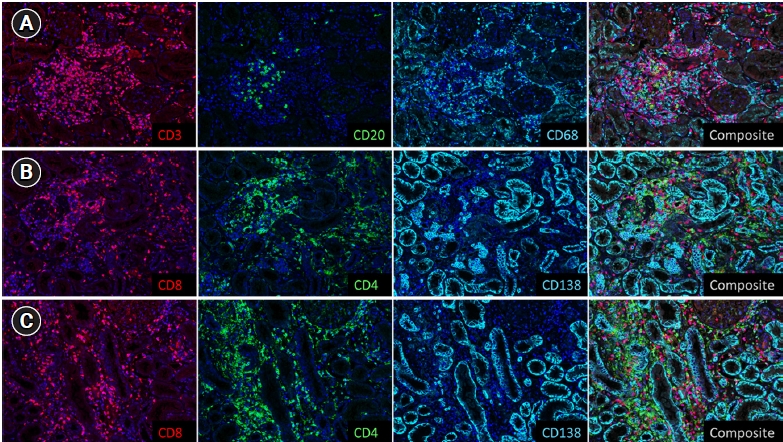
Immunohistochemistry stains showed predominantly CD8+ lymphocytic infiltrate, macrophages, and scattered plasma cells (×100).
(A) The staining results were analyzed using the Vectra imaging system. CD3+ T cells constituted about 80% of the total number of lymphocytes, while CD20+ B cells constituted the remaining 20%. B-lymphocytes formed small aggregates at times. About two-thirds of the T cells were CD8+, while one-third was CD4+. (B, C) Immunohistochemical stains were performed in two different cores of the kidney cortex for better representation. While CD8+ cells were diffusely and evenly distributed throughout the two cores, CD4+ cells tended to form small aggregates, probably in relation to the presence of B-lymphocytes. Scattered CD138+ plasma cells were observed. Overall, the cell types resembled those seen in acute T cell-mediated renal allograft rejection.
Oral prednisolone (PRL) was initiated at 60 mg (1 mg/kg) daily. Omeprazole was replaced with famotidine, a histamine type-2 receptor antagonist, due to the risk of ICI-ATIN potentiation with the continued use of a PPI. Mycophenolate mofetil (MMF) was added in view of inadequate treatment response and steroid-induced dysglycemia. A repeat biopsy at 6 weeks showed improving interstitial inflammation and marginal worsening of interstitial fibrosis. His sCr continued to improve, and PRL and MMF were gradually tapered. The single-agent PD-1 ICI was resumed 5 months after the initial ICI-ATIN episode due to rising tumor markers, with PRL maintained at 10mg daily. The sCr remained stable at 140 μmol/L 10 months after the ICI rechallenge.
Acute tubulointerstitial inflammation is the most common ICI-related kidney injury, either occurring alone or in combination with other glomerular pathologies [1]. The histopathologic features of ICI-ATIN may resemble acute T cell-mediated renal allograft rejection. Interestingly, electron microscopy of our patient’s biopsy revealed subclinical podocyte injury without manifestation of proteinuria. He reported no prior history of diabetes mellitus or use of antivascular endothelial growth factor therapies. The clinical significance of this finding is uncertain, but given reports of podocytopathies with ICI use, ongoing monitoring for the development of proteinuria is warranted.
Notes
Ethical statements
In accordance with our institution’s research policy, a case report involving one or two patients is not defined as research by the Centralized Institutional Review Board and does not require Centralized Institutional Review Board review, as it does not involve the formulation of a research hypothesis that is subsequently investigated prospectively and systematically for publication or presentation. A letter of support from the Research Office of our institution is provided with the submission of our manuscript. Written informed consent was obtained from the patient with permission to publish the case report and accompanying images.
Conflicts of interest
Dr. Joycelyn Lee received an honorarium from Ipsen and Bayer and speaker’s fees from BMS and Ipsen, and received research funding from Bayer. Associate Professor Jason Chon Jun Choo received consultancy fees from Novartis and was awarded research grants from the National Medical Research Council Singapore, Nikko Dento Corporation, and Singhealth Organization. The remaining authors have no conflicts of interests to report, financial or otherwise.
Authors’ contributions
Conceptualization: All authors
Writing–original draft: All authors
Writing–review & editing: All authors
All authors read and approved the final manuscript.
Acknowledgements
We would like to acknowledge Dr. Joe Poh Sheng Yeong for his technical assistance with the images.
