| Kidney Res Clin Pract > Volume 40(4); 2021 > Article |
|
Abstract
Background
Methods
Results
Conclusions
Notes
AuthorsŌĆÖ contributions
Conceptualization: HSB, EMY, KHY, SH, JIS
Data curation, Formal analysis: SY, KHL, HSL, KMJ, KHK, MJL, KHH, HSH, ISH
Investigation: All authors
Methodology: JWL, SJK, TSH, HIC, HGK, MKN, HKC
WritingŌĆōoriginal draft: SY, JYS, MHC, SHK, HC, JSS, JHO, STL, KSK, JHL, YSP
WritingŌĆōreview & editing: SHK, MHC, KML
All authors read and approved the final manuscript.
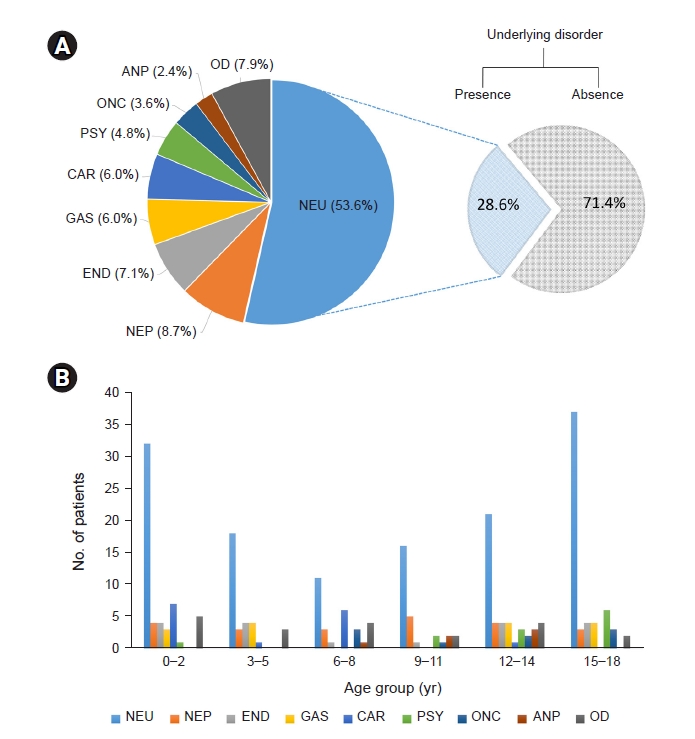
Figure┬Ā2.
Underlying diseases of rhabdomyolysis.
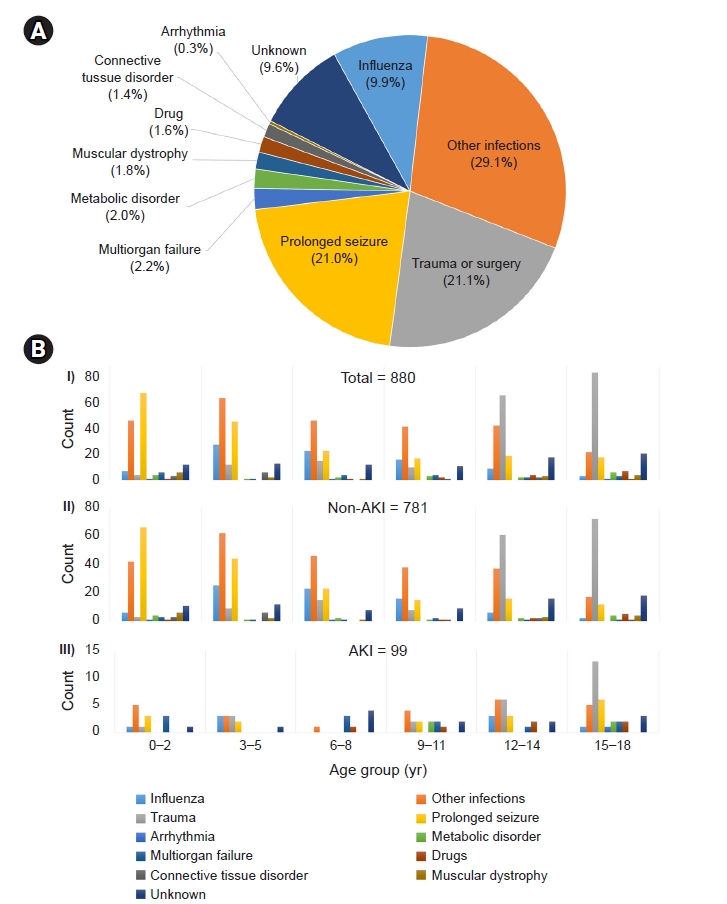
Figure┬Ā3.
Cause of rhabdomyolysis.
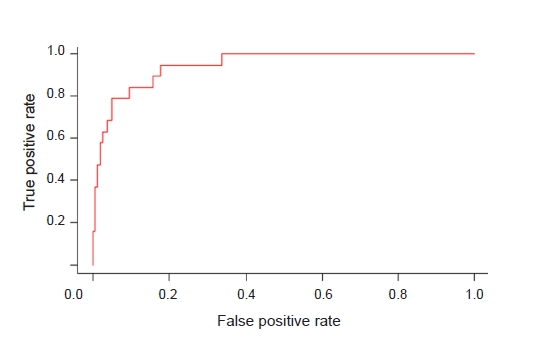
Figure┬Ā4.
Receiver-operating characteristic curve of the multiple logistic regression model for predicting acute kidney injury.
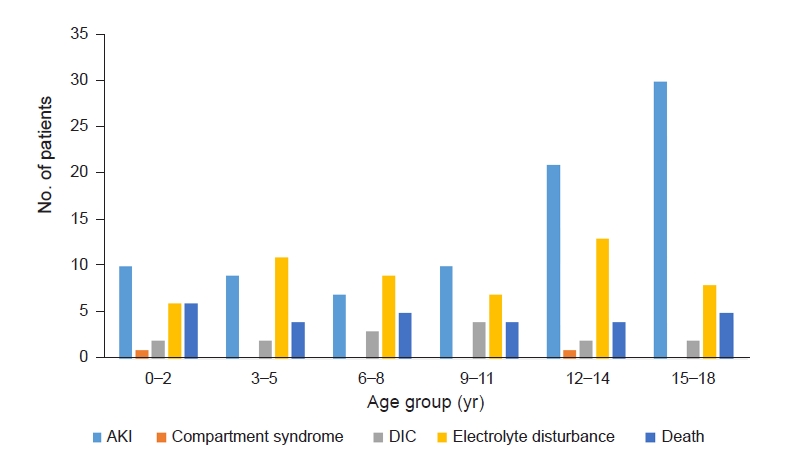
Figure┬Ā5.
Distribution of complications by age in rhabdomyolysis patients.
Table┬Ā1.
| Variable | AKI (n = 99) | Non-AKI (n = 781) | Total (n = 880) | p-value |
|---|---|---|---|---|
| Male sex | 65 (65.7) | 569 (72.9) | 634 (72.0) | 0.17 |
| Age at diagnosis (yr) | 12.2 (6.0ŌĆō15.1) | 8.0 (4.0ŌĆō13.4) | 8.0 (4.0ŌĆō14.0) | <0.001 |
| ŌĆā0ŌĆō2 | 12 (12.1) | 140 (17.9) | 152 (17.3) | |
| ŌĆā3ŌĆō5 | 11 (11.1) | 160 (20.5) | 171 (19.4) | |
| ŌĆā6ŌĆō8 | 9 (9.1) | 119 (15.2) | 128 (14.5) | |
| ŌĆā9ŌĆō11 | 11 (11.1) | 86 (11.0) | 97 (11.0) | |
| ŌĆā12ŌĆō14 | 22 (22.2) | 142 (18.2) | 164 (18.6) | |
| ŌĆā15ŌĆō18 | 34 (34.3) | 134 (17.2) | 168 (19.1) | |
| Height z-score | 0.17 (ŌłÆ1.12 to 1.16) | 0.29 (ŌłÆ0.70 to 1.35) | 0.29 (ŌłÆ0.76 to 1.30) | 0.57 |
| Weight z-score | 0.01 (ŌłÆ1.36 to 0.84) | ŌłÆ0.12 (ŌłÆ1.05 to 0.78) | ŌłÆ0.11 (ŌłÆ1.09 to 0.78) | 0.95 |
| BMI z-scorea (n = 780) | ŌłÆ0.03 (ŌłÆ1.29 to 0.86) | ŌłÆ0.26 (ŌłÆ1.09 to 0.71) | ŌłÆ0.23 (ŌłÆ1.10 to 0.72) | 0.54 |
| BSAb | 1.28 (0.83ŌĆō1.60) | 0.96 (0.70ŌĆō1.39) | 0.97 (0.72ŌĆō1.44) | 0.00 |
| Myalgia or muscle weakness | 41 (41.4) | 301 (38.5) | 342 (38.9) | 0.66 |
| Dark-colored-urine | 20 (20.2) | 103 (13.2) | 123 (14.0) | 0.08 |
| Oliguria, yes | 36 (36.4) | 0 (0) | 36 (4.13) | <0.001 |
| No. of causal factorsc | 0.07 | |||
| ŌĆāSingle | 92 (92.9) | 758 (97.1) | 850 (96.6) | |
| ŌĆāMultiple | 7 (7.1) | 23 (2.9) | 30 (3.4) |
Table┬Ā2.
Data are expressed as number (%), mean ┬▒ standard deviation, or median (interquartile range).
AKI, acute kidney injury; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; CK, creatine kinase; HPF, high-power field; LDH, lactate dehydrogenase; RBC, red blood cell.
Table┬Ā3.
| Variable | OR (95% CI) | p-value |
|---|---|---|
| Age group (yr) | Reference = 3ŌĆō5 yrc | |
| ŌĆā0ŌĆō2 | 0.96 (0.32ŌĆō2.87) | 0.95 |
| ŌĆā6ŌĆō8 | 0.84 (0.24ŌĆō2.89) | 0.78 |
| ŌĆā9ŌĆō12 | 0.72 (0.21ŌĆō2.44) | 0.59 |
| ŌĆā12ŌĆō14 | 1.15 (0.43ŌĆō3.1) | 0.79 |
| ŌĆā15ŌĆō18 | 2.30 (0.89ŌĆō5.93) | 0.09 |
| Presence of underlying disorder | 3.35 (1.91ŌĆō5.88) | <0.001 |
| Multiorgan failure | 6.28 (1.92ŌĆō20.57) | 0.002 |
| Urine occult blooda | Reference = negative | |
| ŌĆāTrace | 0 (0ŌĆōinfinity) | >0.99 |
| ŌĆā1+ | 2.05 (0.78ŌĆō5.38) | 0.15 |
| ŌĆā2+ | 2.69 (1.07ŌĆō6.74) | 0.03 |
| ŌĆā3+ | 4.46 (2.22ŌĆō8.97) | <0.001 |
| ŌĆā4+ | 5.21 (1.53ŌĆō17.81) | 0.008 |
| CK/100a,b | 1 (1.00ŌĆō1.00) | 0.43 |
| AST/100a,b | 1.05 (1.01ŌĆō1.09) | 0.01 |
| Uric acida | 1.23 (1.14ŌĆō1.34) | <0.001 |
| Calciuma | 0.78 (0.65ŌĆō0.94) | 0.008 |
| Phosphorusa | 1.03 (0.89ŌĆō1.19) | 0.73 |
Initial creatinine was included in the multivariable logistic regression analysis to reveal uric acid, phosphorous, and calcium as risk factors for AKI, independent of creatinine.
AKI, acute kidney injury; AST, aspartate aminotransferase; CI, confidence interval; CK, creatine kinase; OR, odds ratio.
Table┬Ā4.
| Variable | AKI (n = 99) | Non-AKI (n = 781) | Total (n = 880) | p-value |
|---|---|---|---|---|
| Short-term outcomes | ||||
| ŌĆāCK recovery time (day) | 7.0 (3.5ŌĆō12.5) | 6.0 (4.0ŌĆō8.0) | 6.0 (4.0ŌĆō9.0) | 0.11 |
| ŌĆāMyoglobin recovery time (day) | 5.0 (3.0ŌĆō11.0) | 5.0 (3.0ŌĆō9.0) | 5.0 (3.0ŌĆō9.0) | 0.44 |
| ŌĆāOliguria recovery time (day) | 2.5 (2.0ŌĆō8.0) | NA | 2.5 (2.0ŌĆō8.0) | 0.005 |
| ŌĆāVolume overload, yes (n=529)a | 24 (33.8) | 3 (0.7) | 27 (5.1) | <0.001 |
| ŌĆāDuration of hospitalization (day) | 18.0 (7.0ŌĆō38.0) | 6.0 (4.0ŌĆō10.0) | 7.0 (4.0ŌĆō12.0) | <0.001 |
| Long-term outcome (n = 522)a | <0.001 | |||
| ŌĆāNone | 39 (82.9) | 460 (96.8) | 499 (95.6) | |
| ŌĆāChronic kidney disease | 3 (6.4) | 0 (0) | 3 (0.6) | |
| ŌĆāRecurrent rhabdomyolysis | 5 (10.5) | 15 (3.2) | 20 (3.8) | |
| Complicationsb | ||||
| ŌĆāNone | 10 (10.1) | 734 (94.0) | 744 (84.6) | <0.001 |
| ŌĆāAKI | 99 (100) | 0 (0) | 99 (11.3) | <0.001 |
| ŌĆāCompartment syndrome | 0 (0) | 2 (0.3) | 2 (0.2) | >0.99 |
| ŌĆāDIC | 12 (12.1) | 3 (0.4) | 15 (1.7) | <0.001 |
| ŌĆāElectrolyte disturbance | 25 (25.3) | 29 (3.7) | 54 (6.1) | <0.001 |
| ŌĆāDeath | 14 (14.1) | 14 (1.8) | 28 (3.2) | <0.001 |
Table┬Ā5.
References
- TOOLS
-
METRICS

- ORCID iDs
-
Sukdong Yoo

https://orcid.org/0000-0003-2365-9134Min Hyun Cho

https://orcid.org/0000-0002-7965-7587Hee Sun Baek

https://orcid.org/0000-0003-0940-360XJi Yeon Song

https://orcid.org/0000-0002-9665-4177Hye Sun Lee

https://orcid.org/0000-0002-2790-2448Eun Mi Yang

https://orcid.org/0000-0001-9410-5855Kee Hwan Yoo

https://orcid.org/0000-0001-6490-4293Su Jin Kim

https://orcid.org/0000-0003-0893-0512Jae Il Shin

https://orcid.org/0000-0003-2326-1820Keum Hwa Lee

https://orcid.org/0000-0002-1511-9587Tae-Sun Ha

https://orcid.org/0000-0002-1249-9342Kyung Mi Jang

https://orcid.org/0000-0002-2226-9268Jung Won Lee

https://orcid.org/0000-0003-1846-3153Kee Hyuck Kim

https://orcid.org/0000-0003-4720-6455Heeyeon Cho

https://orcid.org/0000-0003-3137-6054Mee Jeong Lee

https://orcid.org/0000-0002-7504-1232Jin-Soon Suh

https://orcid.org/0000-0002-6566-6618Kyoung Hee Han

https://orcid.org/0000-0002-6830-7311Hye Sun Hyun

https://orcid.org/0000-0001-8525-1471Il-Soo Ha

https://orcid.org/0000-0001-5428-6209Hae Il Cheong

https://orcid.org/0000-0001-7556-1265Hee Gyung Kang

https://orcid.org/0000-0001-8323-5320Mee Kyung Namgoong

https://orcid.org/0000-0002-8156-8970Hye-Kyung Cho

https://orcid.org/0000-0003-0990-1350Jae-Hyuk Oh

https://orcid.org/0000-0002-8827-7703Sang Taek Lee

https://orcid.org/0000-0003-2570-3290Kyo Sun Kim

https://orcid.org/0000-0001-9322-6508Joo Hoon Lee

https://orcid.org/0000-0001-8010-3605Young Seo Park

https://orcid.org/0000-0001-7653-2036Seong Heon Kim

https://orcid.org/0000-0001-8003-3010 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement 1
Supplement 1 Print
Print















