BK polyomavirus infection following COVID-19 infection in renal transplant recipients: a single-center experience
Article information
Coronavirus disease 2019 (COVID-19) has affected the transplantation community worldwide. Reports of transplant patients acquiring COVID-19 infections are extensive with diverse mortality rates [1]. Follow-up studies of COVID-19 in transplant communities are lacking. There are limited data on the association of the BK polyomavirus (BKPyV) with active COVID-19 infection in kidney transplant recipients (KTRs) [2,3]. Currently, theoretical concerns exist related to graft dysfunction or loss during the post-COVID-19 follow-up period in KTRs. This study aimed to explore the clinical profile, outcomes, and follow-up experiences of KTR patients who developed BKPyV after COVID‐19. This was a single-center retrospective analysis of a study approved by our Institutional Ethical Board (ECR/143/Inst/GJ/2013/RR-19 with application No: EC/App/20Jan21/08) and was conducted in compliance with the Declaration of Helsinki. KTR patients admitted for COVID-19 infection during the study period from June 2020 to December 2020 who developed BKPyV after a positive COVID-19 diagnosis were included. We conducted extended and close monitoring and follow-up of the cohort in the physical, clinical, and psychological domains. Follow-up BKPyV testing was conducted at 1-month after discharge, followed by every 3 months thereafter. Testing also was performed in cases of increasing creatinine.
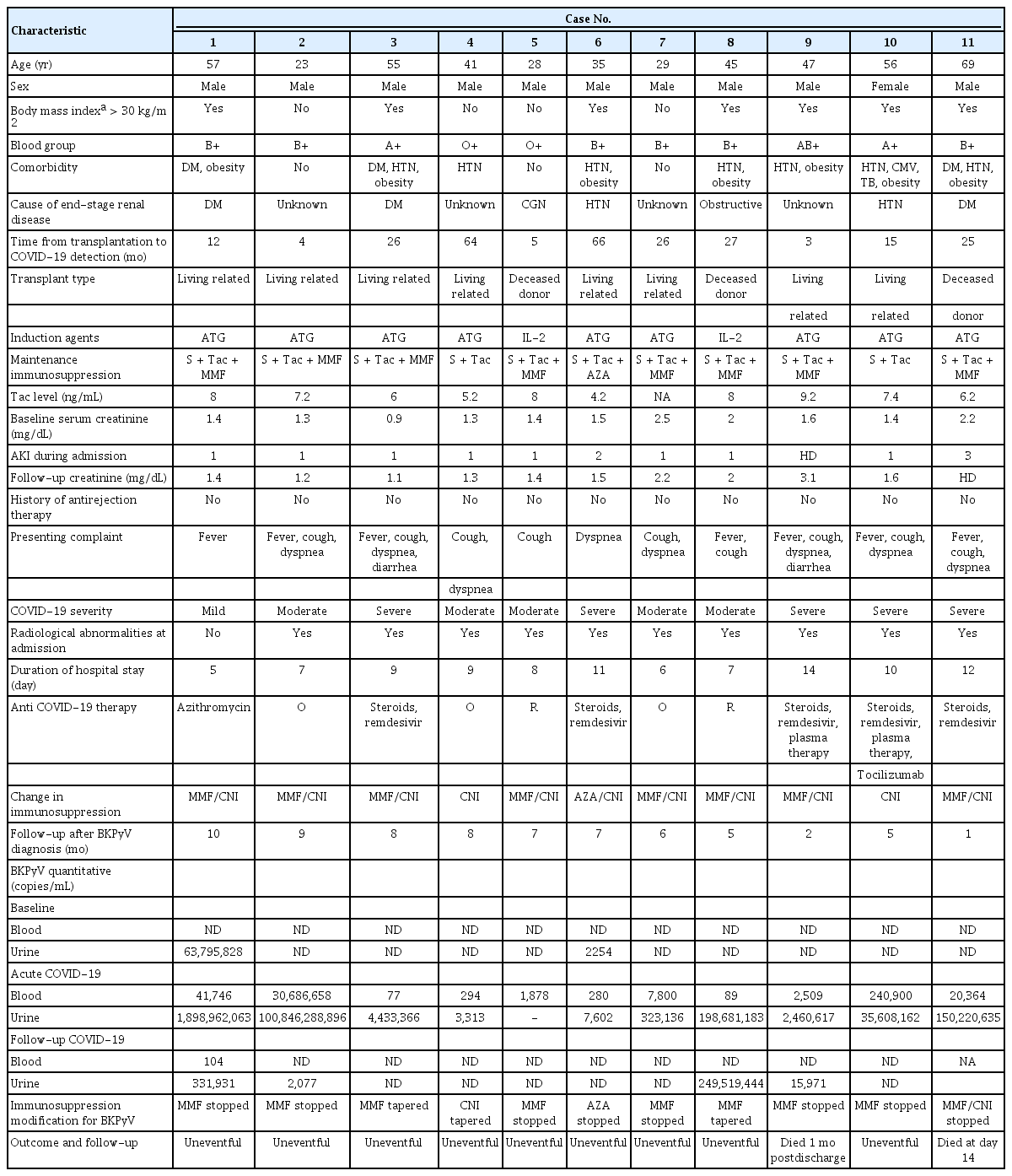
We identified 11 cases of BKPyV after infection in 167 total COVID-19 KTR cases. Table 1 shows the overall summary of the study. The median age of the cohort was 45 years (range, 29–56 years), with male predominance (90.9%). The majority of the cohort had comorbidities (72.7%), underwent live-related-donor transplantation (72.7%), and received thymoglobulin (81.8%) upon admission for COVID-19. The baseline median serum creatinine was 1.44 mg/dL (range, 1.3–1.9 mg/dL). COVID-19 severity was categorized as mild (9%), moderate (45%), and severe cases (46%) [4]. Acute kidney injury was reported in all cases, and acute respiratory distress syndrome developed in 18.2% of KTR patients, with one fatality during COVID-19 admission. Five cases (45.5%) received steroids during acute COVID-19 infection. At baseline, no cases showed BKPyV in the blood. Baseline polymerase chain reaction (PCR) urine testing of the cohort did not detect BKPyV in most cases (81.8%). Table 2 shows the laboratory parameters of the cohort. The median BKPyV blood and urine PCR results during acute COVID-19 infection were 2,509 copies/mL (range, 280–41,746 copies/mL) and 4,433,366 copies/mL (range, 7,602–198,681,183 copies/mL), respectively.
The follow-up period after BKPyV diagnosis was 7 months (range, 5–8 months). BKPyV was detected in the blood during the follow-up period in only one patient. The BKPyV PCR urine values of the cohort were less than those detected in 63.6% of the follow-up cases. No graft loss or graft dysfunction was reported in the cohort. No patient developed sensitization, urine microhematuria, or proteinuria during the follow-up period. Radiological resolution [5] of COVID-19 infection was defined as the absence of any chest radiographic abnormality potentially related to the infection; this type of resolution was seen in 91.6% of KTR cases and resolved after a median of 3 months of follow-up. No multisystemic sequelae were reported. One case was readmitted 1 week after discharge and died due to secondary fungal infection (aspergillosis) after 1 month.
Our report could simply indicate that the natural history and course of BKPyV happened to coincide with COVID-19 infection, and there might be no actual association between the two; however, reactivation of viruses like BKPyV is a high-risk factor for graft loss in transplant patients [6]. BKPyV causes complex changes in immunity and weakens the immune response, which could potentially aggravate the immune/graft injury often present in COVID-19 infection [7]. Elevated levels of inflammatory cytokines in COVID-19 infection can lead to greater transcription of the BKPyV genome [8]. The use of thymoglobulin as an induction agent could have been a confounding factor for BKPyV, but the institutional protocol of using a low dosage of thymoglobulin (1.5 mg/kg) hinders this connection. Moreover, at our center, the incidence of BKPyV in COVID-19 patients was 6.6% (11 of 167 patients), which was higher than the rate reported in normal follow-up or in non-COVID-19 admissions (1.3%). While we were unable to show a definite association of BKPyV with COVID-19 infection, the use of steroids to treat these patients and COVID-19 infection itself are both risk factors for an increase in number of BKPyV in KTRs. Therefore, we suggest screening for BKPyV in COVID-19 patients.
One limitation of this study was its small sample size. To date, this is the largest cohort of KTRs with BKPyV after COVID-19 infection.
In summary, we report BKPyV following COVID-19 with no graft loss during the follow-up period. We suggest screening for BKPyV in all renal transplant patients with active COVID-19 infection (especially in patients with a history of BKPyV and in severe COVID-19 infection) as a safe option to avoid complications.
Notes
Conflicts of interest
All authors have no conflicts of interest to declare.
Authors’ contributions
Conceptualization, Data curation, Formal analysis, Investigation: All authors
Writing–original draft: All authors
Writing–review & editing: All authors
All authors read and approved the final manuscript.