Introduction
Cardiovascular disease (CVD) is a major cause of mortality in patients with end-stage renal disease (ESRD). According to the 2015 Korean registry, 36% of hemodialysis and 39% of peritoneal dialysis patients died of CVD [
1]. The mortality risk for CVD is extremely high in patients with ESRD compared to the general population, even in young chronic dialysis patients. Cardiovascular mortality in patients with ESRD aged Ōēż 25 years is comparable to patients aged 75 to 85 years with normal renal function [
2].
A high incidence of diabetes mellitus, hypertension, and hyperlipidemia in patients with ESRD increases the risk of CVD [
3]. However, these traditional risk factors alone cannot fully explain the increase in CVD incidence and mortality in these patients. Therefore, non-traditional risk factors must be considered. These include uremia-related risk factors such as uremic toxins, inflammation, oxidative stress, and bone mineral diseases [
4]. In addition, fluid overload, infection, bio-incompatibility, and advanced glycation end-products caused by dialysis, may promote proinflammatory cytokine secretion, thereby increasing systemic inflammation and acute-phase responses [
4]. In addition, valvular calcification is common in patients with ESRD, and the risk of infective endocarditis related to frequent vascular access is higher compared to the general population [
3,
5]. Because of these multiple risk factors for CVD in patients with ESRD, the chance of intervention or surgery for treating CVD gradually increases.
The development of renal replacement therapy has improved the survival rate of patients with ESRD. As a result, the demand for cardiac surgery has also increased [
6,
7]. However, since patients with ESRD generally have higher mortality after open heart surgery [
8], chronic dialysis patients may have fewer opportunities for surgical treatment. Therefore, parameters are required to accurately predict postoperative outcomes in patients with ESRD.
EuroSCORE II is widely used to estimate the risk of cardiac surgery and has proven to be a good predictor in the general population [
9ŌĆō
11]. EuroSCORE II is able to predict in-hospital death after cardiac surgery using basic patient information and previous medical history [
10,
11]. The scoring system includes critical preoperative factors such as history of ventricular tachycardia, ventricular fibrillation, acute kidney injury, cardiac massage, and ventilator and intra-aortic balloon pump support, as well as surgery-related risks such as the degree of urgency and thoracic aortic involvement.
Only a few studies have examined outcomes after open heart surgery in patients with ESRD in Korea. Therefore, this study assessed mortality and morbidity after open heart surgery in chronic dialysis patients compared to those in the general population and identified risk factors for postoperative outcomes.
Methods
Subjects
We collected all clinical data from adults aged Ōēź 18 years who underwent open heart surgery between December 2002 and December 2017 in Kyungpook National University Hospital in Daegu, Korea. After excluding those who underwent surgery for cardiac tumors, congenital diseases, and trauma, 2,432 patients were retrospectively analyzed. Among these, we identified 38 patients with ESRD on maintenance dialysis at the time of surgery. We selected a matched control group with normal renal function to assess mortality and morbidity after open heart surgery in the dialysis group. The dialysis group included patients with ESRD undergoing hemodialysis or peritoneal dialysis for at least three months before cardiac surgery. The control group consisted of patients with an estimated glomerular filtration rate (GFR) of Ōēź 60 mL/min/1.73 m2 according to the modification of diet in renal disease GFR equation which had not been diagnosed with any renal disease. We randomly selected 78 patients after matching for age, sex, and diabetes mellitus status. This study was conducted under approval and waiver of informed consent from institutional review board of Kyungpook National University Hospital (KNUH-2018-09-001).
Data collection
We collected data on open heart surgery performed between 2002 and 2017 from the Department of Thoracic and Cardiovascular Surgery at Kyungpook National University Hospital. Baseline characteristics and comorbidities, laboratory data, surgical method, and postoperative outcomes were obtained retrospectively from the medical records, and the outcomes in the dialysis and control groups were compared.
Priority of surgery was classified into three groups. 1) Elective surgery: The medical factors indicate the need for surgery; however, the clinical situation allows for discharge from the hospital with re-admission for surgery at a later date. 2) Urgent: The medical factors require that the patient remain in the hospital before surgery. 3) Emergent: The medical factors relating to the patientŌĆÖs cardiac disease dictate that surgery should be performed within hours to prevent morbidity or mortality.
The type of surgery was divided into four groups including isolated valve procedures, isolated coronary artery bypass grafting (CABG), combined valve and CABG or other procedures, and aortic procedures. The EuroSCORE was calculated according to the age, sex, laboratory data, multiple risk factors, and type of surgery. Risk was classified into three levels (EuroSCORE Ōēż 9%, low-to-moderate risk; EuroSCORE 9ŌĆō25%, high risk; and EuroSCORE > 25%, very high risk). The postoperative outcomes included mortality, duration of hospitalization and ventilator use, and the occurrence of complications. Except for the patients lost to follow-up, mortality after discharge by December 2017 was also investigated.
Statistical analysis
Continuous variables were presented as means ┬▒ standard deviation or medians (interquartile ranges) as appropriate, and categorical variables as the percentage of the sample. Univariate analysis was performed with the Žć2 test for categorical variables and t tests for continuous variables to assess the statistical significance of the differences in patient characteristics between the dialysis and control groups. We used logistic regression analysis to assess the effect of dialysis on each outcome (expressed in terms of odds ratios [ORs]) after adjustment for potentially confounding variables. The potential confounders included sex, age, diabetes mellitus, hypertension, hypercholesterolemia, anemia, and infective endocarditis. Survival curves were estimated using the KaplanŌĆōMeier method and compared using the log-rank test between the two groups. Statistical analysis was performed using SPSS statistics version 23.0 (IBM Corp., Armonk, NY, USA). A P value of Ōēż 0.05 was considered significant.
Results
Table 1 lists the patient characteristics, comorbidities, and laboratory data for the dialysis and control groups. The dialysis and control groups were matched for age, sex, and status of diabetes mellitus. The percentage of males in the dialysis and control groups was 76.3% and 76.5%, respectively (
P = 0.936). There were no significant differences in age (54 ┬▒ 10.7 years vs. 53.6 ┬▒ 10.4 years,
P = 0.849) and percentage of patients aged Ōēź 60 years (26.3% vs. 24.4%,
P = 0.819). At the time of admission, the New York Heart Association (NYHA) class was assessed to determine the symptoms and degree of physical activity (NYHA II [26.3% vs. 56.4%,
P = 0.002], NYHA III [50% vs. 29.5%,
P = 0.031], and NYHA IV [23.7% vs. 14.1%,
P = 0.200]).
Among the various comorbidities, hypertension (97.4% vs. 46.2%, P = 0.000), peripheral vascular disease (10.5% vs. 0%, P = 0.010), anemia (55.3% vs. 9.0%, P = 0.000), and endocarditis (23.7% vs. 5.1%, P = 0.009) were significantly higher in the dialysis group.
There were no significant differences in the incidence of diabetes mellitus, angina, previous myocardial infarction, heart failure, chronic obstructive pulmonary disease, stroke, and liver cirrhosis. The incidence of hypercholesterolemia (5.3% vs. 21.8%,
P = 0.024) was lower in the dialysis group compared to the control group. In addition, a history of smoking, coronary angiography prior to admission, and left ventricular ejection fraction measured using echocardiography were not significantly different between the two groups. Surgical findings including cardiopulmonary bypass time, clamping of aortic time, and use of intra-aortic balloon pump also did not differ between the two groups (
Table 1). EuroSCORE II showed no significant difference between the dialysis and control groups (
Fig. 1).
The priority of surgery determined by the surgeon was presented in
Table 2. The rate of urgent surgery (68.4% vs. 32.1%,
P = 0.000) was significantly higher in the dialysis group, whereas the rate of elective surgery (26.3% vs. 60.3%,
P = 0.001) was significantly higher in the control group. There was no significant difference in the rate of emergent surgery between the two groups. In addition, the type of surgical procedure did not differ between the groups (
Table 2). Crude mortality rates (in-hospital and death after discharge) in both groups are shown in
Table 3. In-hospital mortality rate (18.4% vs. 2.56%,
P = 0.005) and death rate after discharge were significantly higher in the dialysis group. The causes of in-hospital death were as follows: 4 patients died of heart failure, 1 died of stroke, 1 died of brain hemorrhage, and 1 died of bleeding after surgery in the dialysis group, whereas 1 died of heart failure and 1 died of infective endocarditis in the control group. The in-hospital death rates were compared according to the type of procedure and EuroSCORE II. There were no significant differences in mortality by procedure type and the EuroSCORE II between the groups (data not shown).
Deaths were investigated from the time of discharge until December 2017, except in those of follow-up loss (
Table 3). The death rate after discharge was also higher (13.2% vs. 1.3%,
P = 0.014) in the dialysis group (
Table 3): 3 patients died of sepsis, 1 died of gastrointestinal bleeding, and 1 died of unknown causes, while 1 died of stroke in the control group. The median time to death after discharge was 64.9 months in the dialysis group and 31.3 months in the control group.
When postoperative complications were compared, pulmonary edema (26.3% vs. 3.8%,
P = 0.001) and continuous renal replacement therapy (38.5% vs. 2.6%,
P = 0.000) were more frequent in the dialysis group (
Table 4). The hospitalization duration (days) was also longer in the dialysis group (25.8 ┬▒ 28.4 vs. 15.0 ┬▒ 6.3,
P = 0.026). There were no significant differences in other complications. However, the overall complication rates (65.8% vs. 25.6%,
P = 0.000) were significantly higher in the dialysis group (
Table 4). The relative risk of in-hospital mortality for each comorbid condition or type of operation was assessed to confirm the association between each variable and perioperative mortality. Variables with relative risk higher than 2-fold are presented in
Table 5. The relative risk was 16.8 for NYHA class IV, 10.2 for urgent surgery, 4.0 for endocarditis, 4.7 for hypertension, 7.2 for ESRD on dialysis, and 4 for a EuroSCORE II of 9% to 25%. After adjusting for variables including age, sex, hypertension, diabetes, hypercholesterolemia, endocarditis, and peripheral vascular disease, dialysis exhibited the highest relative risk for mortality.
The estimated OR for in-hospital mortality in the dialysis group relative to the control group was 14.7 (
P = 0.033) after adjustment for multiple variables (
Table 6). The total mortality was significantly higher in the dialysis group (adjusted OR, 10.7;
P = 0.029) (
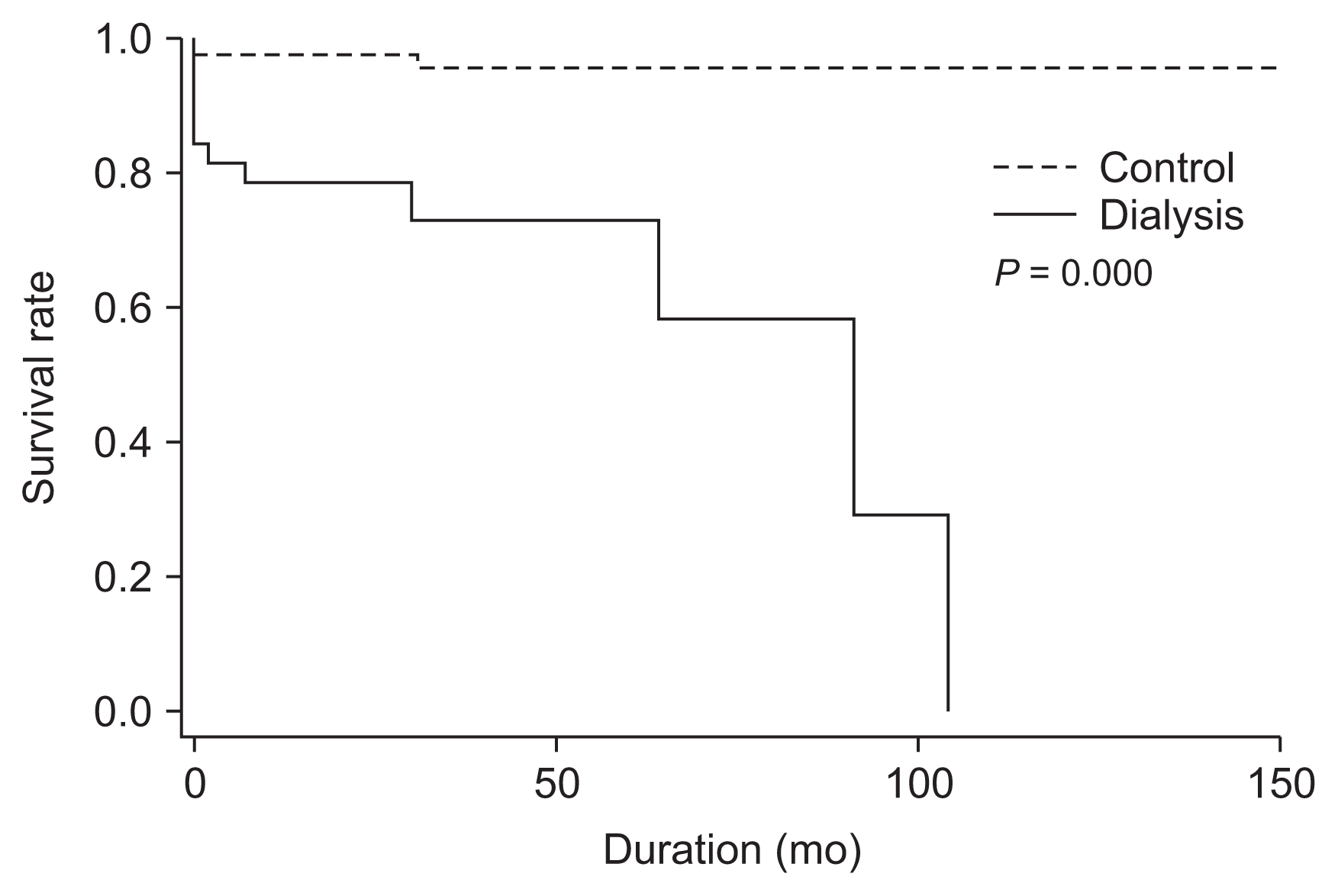
Table 6), while the survival curve using the KaplanŌĆōMeier method also exhibited a significant difference (
P = 0.000) (
Fig. 2). In addition, dialysis increased the risk of complications, including continuous renal replacement therapy (adjusted OR, 36.8;
P = 0.004), hospitalization duration (adjusted OR, 4.6;
P = 0.034), and ventilation duration (adjusted OR, 7.6;
P = 0.022). In addition, the dialysis group showed a 5-fold overall higher risk for complications (adjusted OR 5.7,
P = 0.006) compared to the control group.
Discussion
We compared the outcomes of open heart surgery in patients with ESRD on dialysis with those in a control group with preserved renal function. More comorbidities, higher NYHA class at admission, and greater need for urgent surgery were identified in the dialysis group. The rate of urgent surgery in the dialysis group was extremely high, but the type of procedure and risk predicted by EuroSCORE II was not significantly different. In-hospital mortality was significantly higher in the dialysis group compared to the control group. We estimated the relative risk of mortality through several variables. After adjustment, the relative risk for mortality was the highest in ESRD on dialysis. No significant difference was observed between the dialysis and control groups in terms of EuroSCORE II. EuroSCORE II is used as a predictor of postoperative risk in the general population after open heart surgery, but it did not accurately predict postoperative mortality in the dialysis group [
10,
11]. EuroSCORE II has good predictive value, but the predictive power was limited in chronic dialysis patients. It may be associated with the fact that in chronic dialysis patients, multiple comorbidities mitigate the predictive power of EuroSCORE II.
The dialysis group exhibited a higher risk for mortality after open heart surgery. The causes of death could not be compared directly due to the low mortality rate. In previous studies, chronic dialysis patients showed increased postoperative risk for several reasons. First, chronic dialysis patients are immunocompromised owing to the presence of uremia or diabetes mellitus, steroid use for autoimmune causes of renal failure, and other unknown etiologies [
12]. Second, they possess chronic anemic conditions due to the loss of erythropoietin production, and the presence of anemia is known to be associated with an increase of mortality after surgery [
13ŌĆō
15]. Third, uremia may cause platelet dysfunction and coagulation defects, which may increase bleeding tendencies postoperatively [
16]. Finally, arteriosclerosis, which is common in chronic dialysis patients, may increase the incidence of thromboembolic events and stroke [
17]. In our study, the percentage of patients with diabetes mellitus was similar between the groups. However, the rate of hypertension, anemia, and endocarditis was significantly higher in the dialysis group.
There was no significant difference in postoperative complications except for pulmonary edema or frequency of CRRT use. However, the overall complication rate was significantly higher in the dialysis group. The higher frequency of CRRT therapy is expected during the immediate postoperative period because in chronic dialysis patients, maintenance dialysis is required during the hemodynamically unstable postoperative period.
The mortality rate after discharge was also significantly higher in chronic dialysis patients. We investigated the causes and duration until post-discharge death in both groups. One patient (1.28%) in the control group and five (13.16%) in the dialysis group died during the follow-up period until December 2017. The causes of death after discharge in the dialysis group were stroke, sepsis (n = 3), gastrointestinal bleeding (n = 1), and unknown (n = 1). In the control group, 1 patient died of stroke. The median duration until death after discharge in the dialysis group was longer than in the control group. The higher mortality rate after discharge in the dialysis group seemed to be associated with the higher mortality risk in the ESRD population compared to the normal control rather than the consequences of open heart surgery.
In summary, chronic dialysis patients exhibited higher postoperative mortality, longer hospitalization, and a higher overall rate of complications after open heart surgery compared to the control patients. Dialysis increased the overall mortality risk, especially during the in-hospital period. During the follow-up after discharge, dialysis patients also showed a higher mortality rate compared to the control patients with normal renal function.
In conclusion, chronic dialysis patients have a higher risk of postoperative mortality and morbidity after open heart surgery. Therefore, the benefits of surgical treatment must be balanced against the potential risks of open heart surgery in chronic dialysis patients.





 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print















