Introduction
Recommendations
Infection prevention and control program
Hand hygiene
Isolation strategies
Infection prevention during the dialysis procedure
Control of the source of blood-borne infections
Infection control for vascular access
Water treatment
Waste management
Sterilizing, disinfecting, and cleaning
Laundry care
Environmental control
Vaccination
1. Infection prevention and control program
1.1. Each dialysis unit should develop and implement an infection control program in order to prevent infection and reduce the risk of infection; (IB).
1.2. Dialysis units or centers should establish a rapid response system for emergencies such as epidemics and national disasters related to infectious diseases; (IB, IC).
-
1.3. The following activities should be carried out according to the rules and policies of each institution; (IB).
The planning, implementation and surveillance of infection control measures to protect the health of dialysis staff
Infection control education and training for dialysis staff
Sterilizing, disinfecting, and cleaning equipment and supplies
Environmental control (disinfecting and cleaning)
Initiation and maintenance of dialysis, control of the source of blood-borne infections, infection control for vascular access, water treatment, and vaccination
Basic protective measures such as hand hygiene, isolation precautions, and aseptic techniques
Operating a healthcare-associated infection surveillance system
Management of outbreaks
2. Hand hygiene
2.1. Dialysis staff should perform hand hygiene in an appropriate manner when hand hygiene is required during dialysis.
2.2. The situations where hand hygiene is required are as follows; (IB):
2.2.1. Before touching a patient (e.g., measurement of vital signs, evaluation of vascular access)
-
2.2.2. Before performing a clean or aseptic procedure
Before intervention (e.g., dialysis preparation)Before drug administrationBefore management of an area with broken skin integrity (e.g., catheter dressing)Before an invasive procedure (e.g., cannulation of vascular access or vein puncture) 2.2.3. After body fluid exposure risk (e.g., hemostasis after dialysis therapy)
2.2.4. After touching a patient (e.g., measurement of vital signs)
2.2.5. After touching a patient’s surroundings (e.g., finishing dialysis therapy)
2.3. Hemodialysis patients, as well as their families and visitors, should also perform hand hygiene in an appropriate manner when entering the dialysis room.
Background
Hand hygiene in the hemodialysis process
3. Isolation strategies
3.1. Quarantine measures may be taken if necessary to prevent the transmission of infections from patients with suspected or confirmed infections.
3.2. Patients with acute respiratory infection symptoms should be instructed to wear surgical masks and follow proper cough etiquette when entering the dialysis room.
3.3. Standard precautions should be applied when dealing with all suspected or confirmed infections in patients; (IA). Depending on the mode of transmission (contact, droplet, and/or airborne), additional precautions should be taken.
3.4. A patient with an infectious disease for which contact or droplet precautions are required should either receive dialysis therapy in a single-occupancy isolation room, by the cohort isolation method, or be last in the order after others have completed their dialysis therapy.
3.5. A patient with an infectious disease for which airborne precautions are required can receive dialysis in either a negative-pressure room or a single-occupancy isolation room.
3.6. Inter-hospital and intra-hospital transfer of patients who require isolation care should be limited to absolutely necessary cases; (II).
Background
Basic principles of isolation according to the mode of transmission of the pathogen
4. Infection prevention during the dialysis procedure
4.1. The catheter exit site or cannulation site of an arteriovenous fistula or arteriovenous graft should be examined for the absence of infection before the bloodstream is accessed at each hemodialysis session; (IB).
4.2. Hand hygiene and aseptic techniques should be followed throughout all dialysis procedures; (IB).
4.3. The use of common supplies between patients is prohibited.
4.4. Injections should be prepared in a room or area separate from the patient treatment area and administered to patients after their identities have been checked; (IB).
4.5. For the preparation of multi-dose injections (e.g., heparin), each dose should be weighed in a separate syringe for each patient in the preparation room, and multi-dose injection vials should not be taken to the patient treatment area.
Infection control measures during the dialysis procedure
Principles of using multi-dose injections
5. Control of the source of blood-borne infections
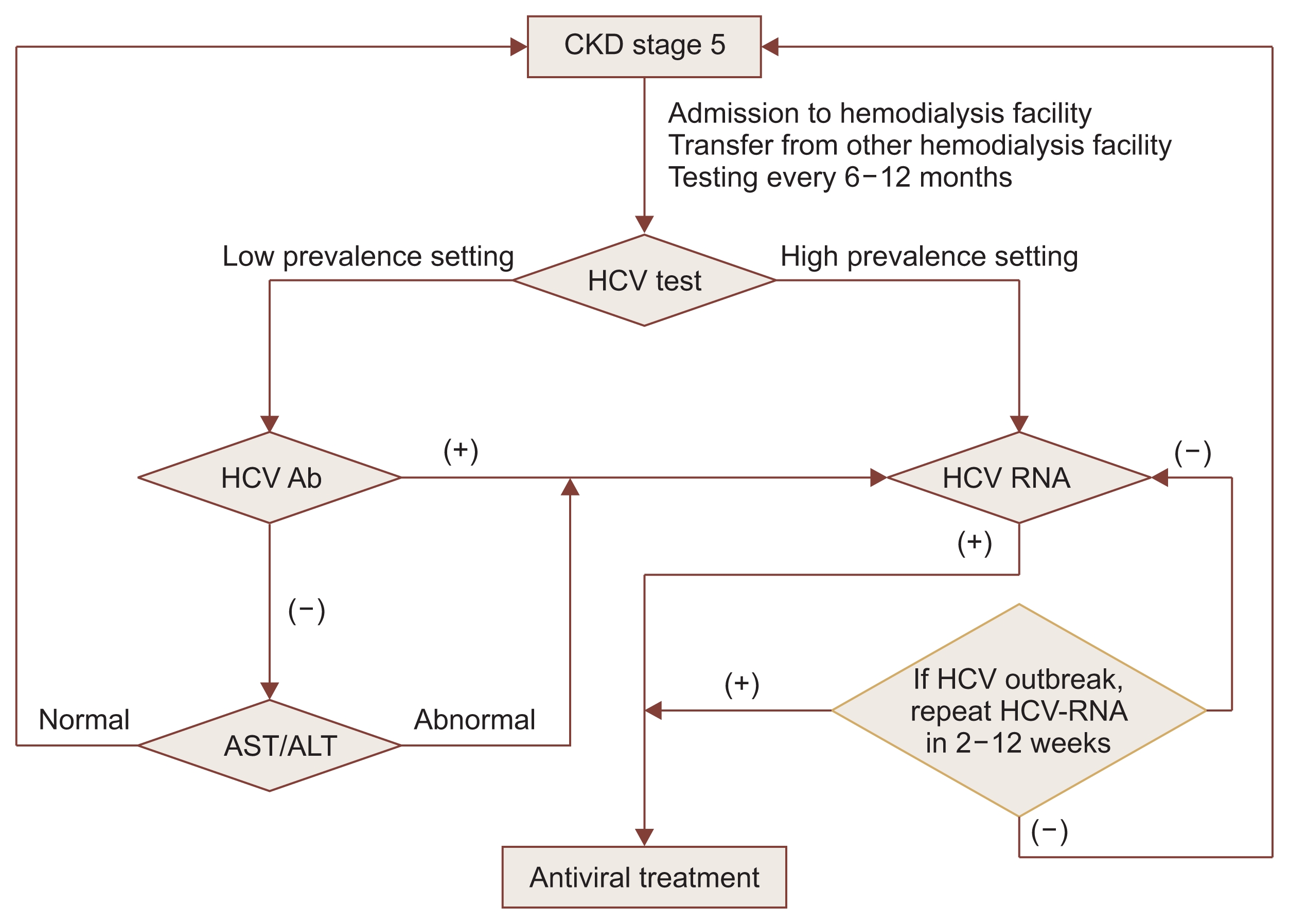
5.1. All hemodialysis patients should undergo routine screening for hepatitis B and hepatitis C infections; (IB).
5.2. Patients at risk for hepatitis B infection should be vaccinated against hepatitis B virus (HBV).
5.3. Patients who are hepatitis B surface antigen (HB-sAg) positive should be dialyzed with separate machines, equipment, instruments, and supplies; (IB).
5.4. Patients with hepatitis C and human immuno-deficiency virus (HIV) infections are not recommended to be isolated from other patients or dialyzed separately on dedicated machines if standard precautions are followed; (IB).
Background
HBV isolation and precautions
HCV and HIV precautions
6. Infection control for vascular access
6.1. Temporary catheters should be kept for a minimum period and removed as soon as possible; (IA).
6.2. The catheter exit site and catheter hub should be examined for the absence of infection at each hemodialysis session when a patient uses a catheter for dialysis; (IB).
6.3. The employee should wear a mask and sterile gloves when the catheter is being manipulated; (IB).
6.4. The vascular access site should be washed with antibacterial soap and water before cannulation when an arteriovenous access is used for dialysis.
6.5. The employee should follow aseptic techniques for vascular cannulation; (IB).
Insertion of a hemodialysis catheter
Exit site care when a hemodialysis catheter is used
7. Water treatment
7.1. The use of ultrapure water is considered vital during hemodialysis, especially in the case of high-flux dialysis or hemodiafiltration (HDF).
7.2. The water produced for dialysis should be tested regularly for bacteriological and chemical contaminants. When the results of assays exceed acceptable levels, corrective measures should be taken promptly to reduce the levels of bacteria/ chemicals.
7.3. Bacteriological assays of water should initially be performed weekly to validate the adequacy of the dialysis machine disinfection process, and should then be performed monthly once the process has been established.
7.4. Chemical tests of water should be performed annually.
8. Waste management
8.1. The materials used in the hemodialysis process, such as dialysis membranes, lines, or needles, should be treated as biomedical waste, placed in a special container, and handled by a waste disposal contractor.
8.2. Used hemodialysis fluid is not considered biomedical waste since it does not make direct contact with blood.
9. Sterilizing, disinfecting, and cleaning
10. Laundry care
11. Environmental control
11.1. The guidelines for environmental infection control in each healthcare facility should be followed so that all areas occupied by patients, employees, and visitors are kept clean and safe.
11.2. The hemodialysis machine should be rinsed after each dialysis session, and the interior of the dialysis machine should be disinfected at the end of the day; (IB).
11.3. The waste treatment area (cleaning room and sewage room) should be separate from the patient treatment area, storage room, and clean area.



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print
















