Fatty liver associated with metabolic derangement in patients with chronic kidney disease: A controlled attenuation parameter study
Article information
Abstract
Background
Hepatic steatosis measured with controlled attenuation parameter (CAP) using transient elastography predicts metabolic syndrome in the general population. We investigated whether CAP predicted metabolic syndrome in chronic kidney disease patients.
Methods
CAP was measured with transient elastography in 465 predialysis chronic kidney disease patients (mean age, 57.5 years).
Results
The median CAP value was 239 (202–274) dB/m. In 195 (41.9%) patients with metabolic syndrome, diabetes mellitus was more prevalent (105 [53.8%] vs. 71 [26.3%], P < 0.001), with significantly increased urine albumin-to-creatinine ratio (184 [38–706] vs. 56 [16–408] mg/g Cr, P = 0.003), high sensitivity C-reactive protein levels (5.4 [1.4–28.2] vs. 1.7 [0.6–9.9] mg/L, P < 0.001), and CAP (248 [210–302] vs. 226 [196–259] dB/m, P < 0.001). In multiple linear regression analysis, CAP was independently related to body mass index (β = 0.742, P < 0.001), triglyceride levels (β = 2.034, P < 0.001), estimated glomerular filtration rate (β = 0.316, P = 0.001), serum albumin (β = 1.386, P < 0.001), alanine aminotransferase (β = 0.064, P = 0.029), and total bilirubin (β = −0.881, P = 0.009). In multiple logistic regression analysis, increased CAP was independently associated with increased metabolic syndrome risk (per 10 dB/m increase; odds ratio, 1.093; 95% confidence interval, 1.009–1.183; P = 0.029) even after adjusting for multiple confounding factors.
Conclusion
Increased CAP measured with transient elastography significantly correlated with and could predict increased metabolic syndrome risk in chronic kidney disease patients.
Introduction
Metabolic syndrome (MS), comprising hypertension, hypertriglyceridemia, hyperglycemia, low high-density lipoprotein cholesterol (HDL-C) levels, and central obesity, is closely correlated with insulin resistance, chronic inflammation, and cardiovascular disease development [1]. Several studies reported that MS is associated with chronic kidney disease (CKD) or albuminuria [2,3], and that patients with MS have increased all-cause and cardiovascular mortality in both CKD and the general populations [4]. Regarding the relationship between MS and CKD, several epidemiologic studies demonstrate that hyperglycemia, hypertension, low HDL-C levels, and hypertriglyceridemia were major risk factors for CKD development and progression. However, waist circumference, body mass index (BMI), and obesity, important signs of insulin resistance of MS, are confusing factors, especially in CKD patients.
Obese populations with renal impairment showed reverse epidemiology compared with the general population [5]. This paradox is not explained by traditional pathophysiologic mechanisms; thus, investigation of factors such as sarcopenic obesity [6], waist-to-hip ratio [7], or metabolically healthy obesity [8] is needed. Among these surrogate findings of metabolic derangements, interest in the liver as a phenotype of sarcopenia and central obesity has been increasing in the last decade [9]. Recent observations suggest that fatty liver or steatohepatitis, are a surrogate marker for MS. In addition, non-alcoholic fatty liver disease (NAFLD) is an extremely common liver disease accompanied by metabolic derangements and is considered a hepatic manifestations of MS [10]. NAFLD is strongly associated with insulin resistance caused by low physical activity and increased fat mass [11].
Transient elastography (FibroScan; Echosens, Paris, France) was originally invented for non-invasive detection and assessing liver cirrhosis severity [12]. Transient elastography has recently been used for evaluating liver elasticity and viscosity with ultrasonic signals, known as the controlled attenuation parameter (CAP). Accumulating evidences suggest that CAP could estimate the degree of hepatic steatosis and be a substitute for invasive liver biopsies [13]. Moreover, previous studies showed that CAP is useful in assessing the severity of NAFLD and is closely associated with MS in the general populations [14].
Therefore, this study aimed to elucidate whether CAP measurement using transient elastography has a predictive role in assessing metabolic derangements in CKD patients.
Methods
Ethics statement
The study was carried out in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Yonsei University Health System (YUHS) Clinical Trial Center.
Subjects
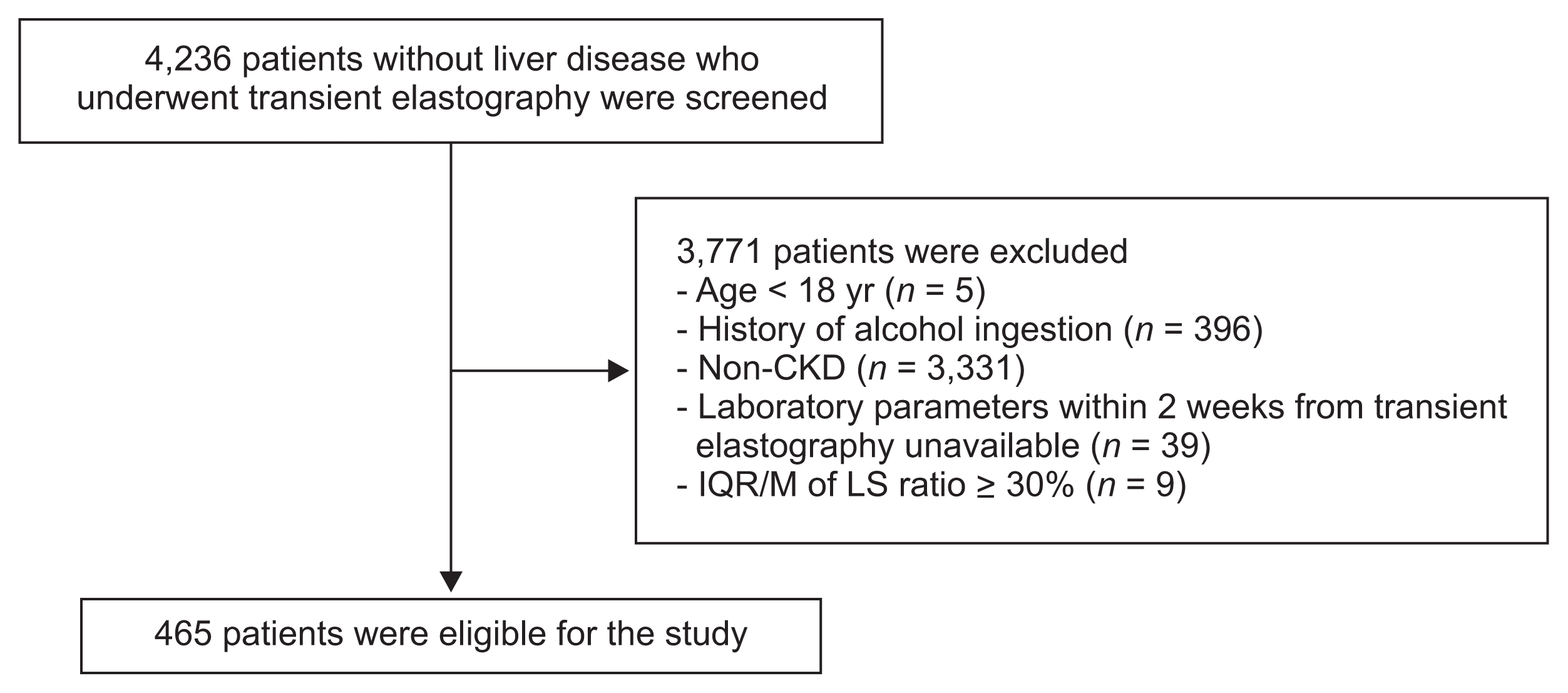
All pre-dialysis CKD patients without liver disease who underwent transient elastography at YUHS were initially screened for enrollment between November 2011 and December 2014 in this observational study. Patients under 18 years old were excluded. The exclusion criteria included the presence of hepatitis B surface antigen, hepatitis C virus antibodies, concomitant or previous history of liver cirrhosis, alcoholic hepatitis, alcoholic liver disease, drug induced hepatitis, fulminant hepatitis, autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, hepatocellular carcinoma, biliary tract cancer, gallbladder cancer, any cancer with metastasis to the liver, or type 1 hepatorenal syndrome. Patients whose MS parameters were unavailable were also excluded. Finally, 465 CKD patients were analyzed (Fig. 1).
Clinical and biochemical data collection
Demographic and clinical data were collected from the registered database at YUHS. BMI was calculated as weight divided by square of height (kg/m2). Diabetes mellitus and hypertension were defined as previous described [15,16]. CKD stage was defined by the Kidney Disease Improving Global Outcomes 2012 guideline [17], and estimated glomerular filtration rate (eGFR) was calculated by the CKD epidemiology collaboration equation [18]. Biochemical data including urine albumin-to-creatinine ratio (UACR) was analyzed before or after 6 months at the time of transient elastography. Serum total cholesterol, HDL-C, low-density lipoprotein cholesterol, and triglyceride levels were measured by enzymatic colorimetry using an autoanalyzer (Hitachi 7150; Hitachi Ltd, Tokyo, Japan), and high-sensitivity C-reactive protein (hs-CRP) levels were determined by a latex-enhanced immunonephelometric method using a BNII analyzer (Dade Behring, Newark, DE, USA).
Diagnosis of metabolic syndrome
The National Cholesterol Education Program (Adult Treatment Panel [ATP] III) [19] bases the diagnosis of MS on the presence of three or more of the following: 1) systolic blood pressure ≥ 130 mmHg and/or diastolic blood pressure ≥ 85 mmHg; 2) triglyceride levels ≥ 150 mg/dL, 3) HDL-C levels < 40 mg/dL in men or < 50 mg/dL in women; 4) fasting plasma glucose levels ≥ 110 mg/dL; and 5) abdominal obesity. We based abdominal obesity on BMI rather than on waist circumference because of the participants with a BMI ≥ 25 kg/m2 classified centrally obese in accordance with the Western Pacific Region of the World Health Organization (WPRO) criteria [20]. The modified ATP III definition used in this study has been determined to be more accurate than the WPRO criteria for the diagnosis of MS in Asians [21].
Liver stiffness and controlled attenuation parameter measurements
Liver stiffness (LS) and CAP measurements were performed by transient elastography on the right lobe of the liver through the intercostal spaces while patients were lying in the dorsal decubitus position with the right arm in maximal abduction. The final LS result was expressed in kPa and was the median value of 10 measurements performed between 25 and 65 mm depth. Only result with 10 valid shots and an interquartile range divided by median LS ratio < 30% were included [12].
The principle of CAP measurement has been described previously [22]. In brief, the CAP measures reflected ultrasonic attenuations at 3.5 MHz using signals acquired by transient elastography. The final CAP was computed only when the associated LS measurement was valid and using the same signals as the one used to measure LS. Therefore, both LS and CAP were obtained simultaneously and in the same volume of liver parenchyma. The final CAP value was presented by the median of individual CAP values and was expressed in dB/m [14].
Statistical analysis
Continuous variables were expressed as mean ± standard deviation or median (range), while categorical variables were expressed as a number (percentage). Normality of distribution was ascertained with the Kolmogorov-Smirnov test. To compare differences between patients with and without MS, Student’s t-test or the Mann-Whitney U-test were used for continuous variables, and the chi-square test or the Fisher’s exact test were used for categorical variables. Univariate and multiple linear regression analyses were performed to determine significant factors associated with LS and CAP. Regarding variable distribution, UACR, hs-CRP, and LS were used with natural log values for linear regression analysis. The variables with P-values < 0.05 with univariate linear regression analysis were included in multiple linear regression analysis. The independent predictive role of CAP for the presence of MS was confirmed through logistic regression analysis, which included significant variables when comparing baseline characteristics according to the presence of MS and multiple linear regression analysis. We further modified the above logistic regression analyses by using a restricted cubic spline model to illustrate systemic relations between the degree of CAP values and the risk of MS. The statistical significance of the mean CAP across the number of MS components was verified with the Trend test. Statistical analysis was performed with IBM SPSS software for Windows version 23.0 (IBM Corporation, Armonk, NY, USA). P-values < 0.05 were considered significant.
Results
Baseline characteristics according to the presence of metabolic syndrome
The baseline characteristics are shown in Table 1. The mean age of the study population was 57.5 ± 17.0 years, and 225 (48.4%) patients were men. There were 265 (57.0%), 131 (28.2%), 43 (9.2%), 16 (3.4%), and 10 (2.2%) patients with stage 1 to 5 CKD, respectively. The mean eGFR was 87.9 ± 28.3 mL/min/1.73 m2, and the median CAP was 239 dB/m. MS was present in 195 (41.9%) patients. Patients were divided into two groups according to the presence of MS. MS group had more diabetes mellitus (105 [53.8%] vs. 71 [26.3%], P < 0.001), significantly higher UACR (184 [38–706] vs. 56 [16–408] mg/g Cr, P = 0.003), hs-CRP levels (5.4 [1.4–28.2] vs. 1.7 [0.6–9.9] mg/L, P < 0.001), LS (5.6 [4.6–8.6] vs. 4.9 [4.1–7.1] kPa, P = 0.003), and CAP (248 [210–302] vs. 226 [196–259] dB/m, P < 0.001) compared with non-MS group.
Linear regression analyses performed with liver stiffness or controlled attenuation parameter
Univariate linear regression analysis was performed to investigate the association between clinical and biochemical variables with the value measured with transient elastography (Table 2). Fasting glucose, serum creatinine, log UACR, aspartate aminotransferase (AST), alanine transaminase (ALT), total bilirubin, and log hs-CRP levels were positively correlated with log LS, while cholesterol, HDL-C, and serum albumin levels were negatively correlated with log LS. BMI, diastolic blood pressure, the presence of diabetes mellitus, fasting glucose, cholesterol, log triglyceride, eGFR, serum albumin, and ALT levels were positively correlated with CAP, while age, BUM, and total bilirubin levels were negatively correlated with CAP values.

Univariate linear regression analyses for the association of liver stiffness and controlled attenuation parameter with variables
Multiple linear regression analysis was also performed to clarify the independent demographic and biochemical variables with LS or CAP values in patients with CKD (Table 3). The analysis revealed that log LS was independently associated with log UACR (β = 0.063, P = 0.026) and total bilirubin levels (β = 0.309, P < 0.001), while CAP was independently associated with BMI (β = 0.742, P < 0.001), log triglyceride (β = 2.034, P < 0.001), eGFR (β = 0.316, P = 0.001), serum albumin (β = 1.386, P < 0.001), ALT (β = 0.064, P = 0.029), and total bilirubin levels (β = −0.881, P = 0.009).
Controlled attenuation parameter is an independent risk factor for the presence of metabolic syndrome in patients with chronic kidney disease
Logistic regression analysis was performed to investigate the predictive impact of CAP on the prevalence of MS in CKD patients (Table 4). CAP was significantly associated with the prevalence of MS in a crude model (CAP, 10 dB/m increase; odds ratio [OR], 1.101; 95% confidence interval [CI], 1.060–1.143; P < 0.001; data not shown). In addition, a significant association remained after adjustment for not only liver function-related factors but also traditional risk factors including age, sex, diabetes mellitus, hypertension, total bilirubin levels, serum albumin levels, eGFR, UACR, hs-CRP, and HDL-C levels (CAP, 10 dB/m increase; OR, 1.093; 95% CI, 1.009–1.183; P=0.029). On the other hand, LS did not show a significant correlation with the presence of MS in CKD patients. The association between CAP groups which were divided into five groups, and full-adjusted odds ratio was constructed and described as cubic spline curves in Fig. 2. As shown, the OR is increased from around 3rd group, then the statistical significance is appears around 4th group. In addition, the mean value of CAP linearly increased with the number of MS components, and the trend showed significant statistic power (P for trend < 0.001, Fig. 3).

Controlled attenuation parameter as an independent predictor of metabolic syndrome in patients with chronic kidney disease
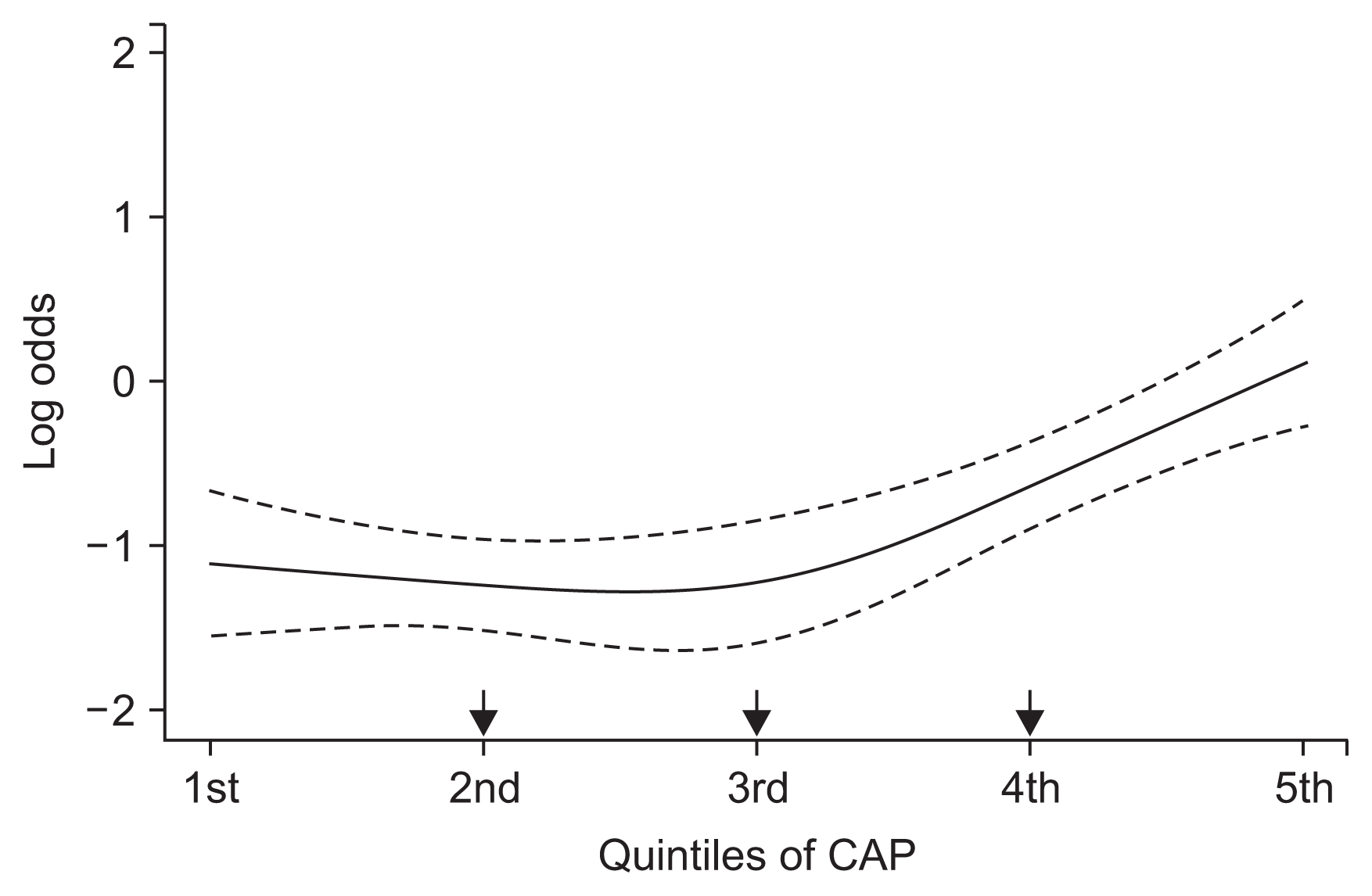
A cubic spline curves for the presence of metabolic syndrome according to the degree of CAP values in patients with chronic kidney disease
The models were adjusted for age, gender, history of diabetes mellitus and hypertension, total bilirubin, serum albumin, eGFR, UACR, hs-CRP, and HDL-C. The solid line represents the log odds of metabolic syndrome according to CAP, and the dotted line representes 95% CI.
CAP, controlled attenuation parameter; CI, confidence interval; eGFR, estimated glomerular filtration rate; HDL-C, high-density lipoprotein cholesterol; hs-CRP, high sensitivity C-reactive protein; UACR, urine albumin-to-creatinine ratio.
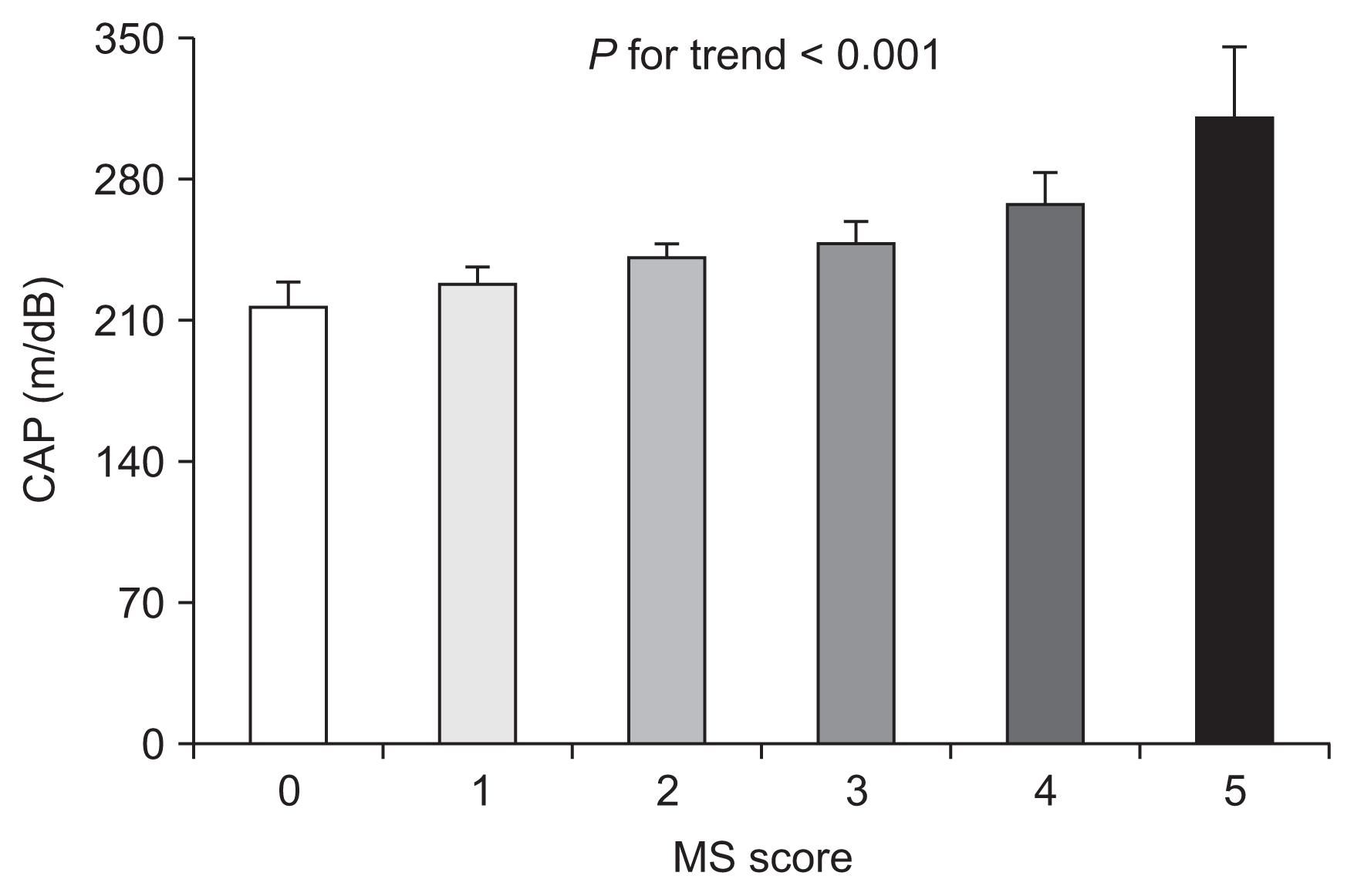
Controlled attenuation parameter according to the number of metabolic syndrome (MS) components
Each bar represents the mean and 95% confidence interval. Controlled attenuation parameter values increased in parallel with the number of metabolic syndrome components (P < 0.001).
CAP, controlled attenuation parameter.
Body mass index and triglyceride levels are the main factors affecting the predictive role of controlled attenuation parameter in diagnosing metabolic syndrome
To investigate the main factors affecting the predictive power of CAP in diagnosing MS, the proportion of diagnosed MS components according to the dichotomized CAP is presented (Fig. 4). High and low CAP groups were generated by the median value of CAP score. Among the five diagnostic criteria of MS, BMI (27.7% vs. 72.3%, P < 0.001) and triglyceride levels (32.5% vs. 67.5%, P < 0.001) were significantly more prevalent in the higher-CAP compared with the lower-CAP group, but fasting plasma glucose levels, HDL-C levels, and systolic blood pressure were not significantly different between groups.
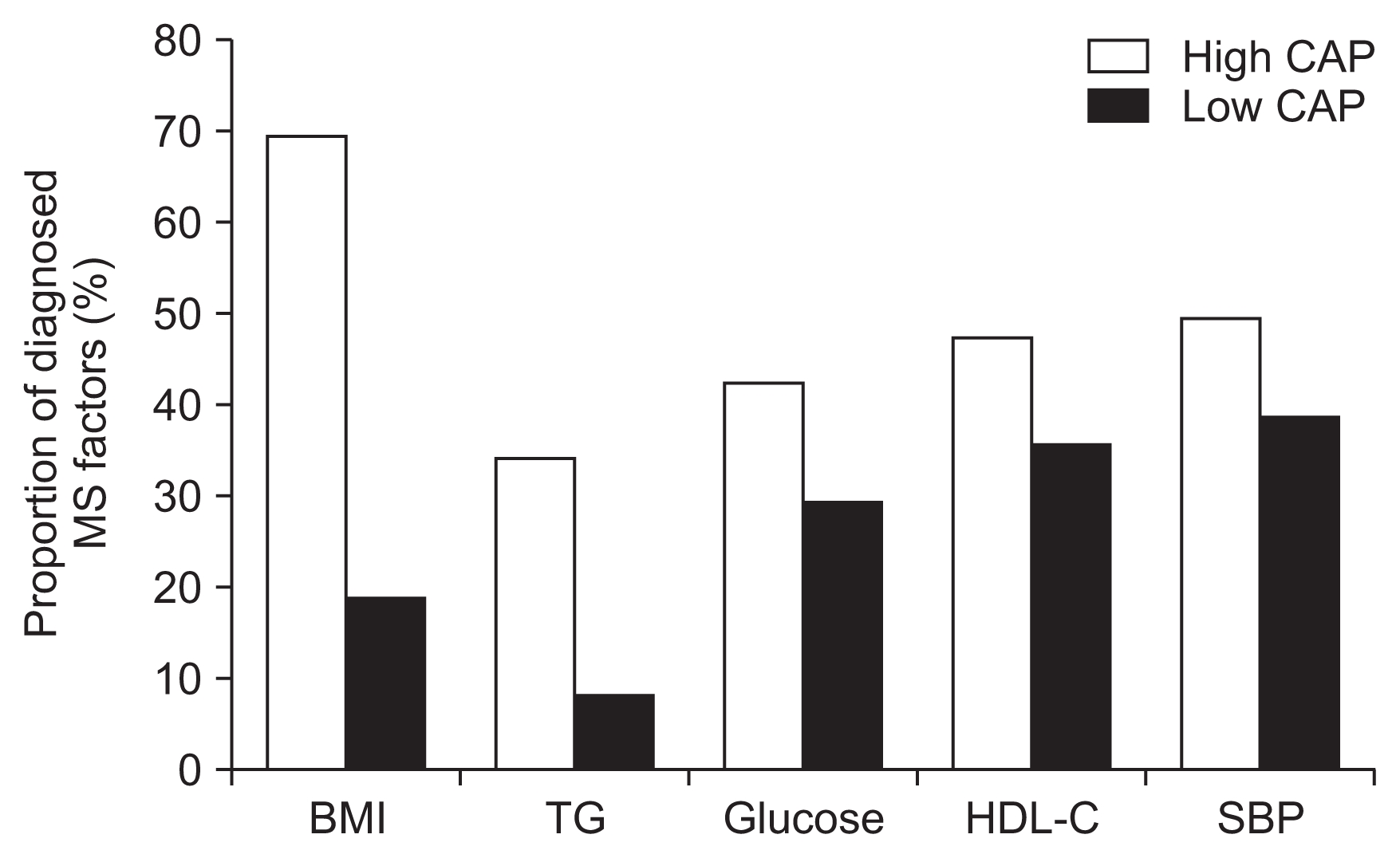
The proportions of metabolic factors which satisfy the criteria of metabolic syndrome (MS) according to the dichotomized CAP
Among the five diagnostic criteria of MS, BMI (27.7% vs. 72.3%, P < 0.001) and TG (32.5% vs. 67.5%, P < 0.001) showed significant higher proportions in the higher CAP group than the lower CAP group, but fasting plasma glucose, HDL-C, and SBP did not show differences between the two groups.
BMI, body mass index; CAP, controlled attenuation parameter; HDL-C, high-density lipoprotein cholesterol; SBP, systolic blood pressure; TG, triglyceride.
Discussion
In the present study, we found that the CAP value was independently associated with the presence of MS in pre-dialysis CKD patients. The liver and muscles are target organs for insulin action. Recently, the role of the liver in insulin resistance is being investigated; several observational studies suggest that NAFLD is closely associated with insulin resistance and sarcopenia [23]. Hitherto, the underlying mechanisms for an association between NAFLD and sarcopenia are not fully elucidated. Previous studies proposed that hepatic steatosis was strongly correlated with insulin resistance, mitochondrial dysfunction, and consequent muscle breakdown [24]. In particular, several adipokines produced by adipose tissues are reported to be involved in modulating insulin action in metabolic tissues such as the muscles and liver [25]. Furthermore, patients with chronic liver disease or cirrhosis reportedly have lower levels of insulin growth factor-1 and increased levels of various cytokines such as tumor necrosis factor alpha and transforming growth factor beta, which are significantly associated with sarcopenia [26]. In a previous study, sarcopenia was commonly reported in CKD patients and was related to protein-energy wasting, inflammation, and cardiovascular mortality in them [6,27]. In our study, CAP values as a marker for hepatic steatosis were significantly higher in CKD patients with MS in accordance with previous studies performed on patients without kidney abnormalities.
Studies on the association between obesity and metabolic risk factors have shown conflicting results in CKD patients. Although an inverse association between BMI and survival rates is reported in CKD patients including dialysis patients [28,29], several studies show that central obesity assessed through anthropometric measurements is associated with a higher risk of cardiovascular mortality in patients with CKD as well as dialysis patients [30]. These data suggested that metabolic abnormalities affected by visceral adiposity might be an important risk factor for cardiovascular mortality and morbidity. Accumulation of visceral fat leads to increased excretion of pro-inflammatory cytokines that may stimulate aggravation of hepatic steatosis and have a catabolic effect on the skeletal muscles [31]. Since CKD leads to metabolic abnormalities such as impaired glucose and protein metabolism, increased oxidative stress, and systemic inflammation, MS in CKD patients is frequently observed and is also considered a non-traditional risk factor for cardiovascular outcomes. The present study suggested that measurement of hepatic steatosis could help to detect MS in CKD patients.
Transient elastography is a newly invented diagnostic tool that is non-invasive, painless, easy to perform, and has superior reliability for pathologic diagnosis [32]. Because of the unique role of the liver in the regulation of metabolic derangement, the use of transient elastography has been expanded to evaluate NAFLD and MS [9]. CAP is calculated by using the velocity and intensity of reflected ultrasonic signals; it reveals the stiffness, viscosity, and elasticity of the liver; thus, CAP could measure hepatic steatosis.
The CAP score, distinct from the LS score obtained through transient elastography, has been recently established and determined as a reliable method to detect hepatic steatosis [13,14,22]. In addition, a large prospective study revealed that CAP was significantly correlated with traditional metabolic risk factors such as age, sex, and BMI as well as MS itself [14]. One previous study already revealed the usefulness of CAP in CKD patients, showing that hepatic steatosis measured with CAP was significantly correlated with serum creatinine and hs-CRP levels [33]. The present study also demonstrated that LS was closely correlated with liver function parameters such as serum albumin and prothrombin time [12]. CAP was significantly increased in CKD patients with MS, and CAP was independently associated with BMI and triglyceride levels in multiple linear regression analysis. In addition, increased CAP was independently associated with the presence of MS in the CKD population after adjustment for multiple factors, including liver function-related and traditional risk factors. Further analysis indicated that the significant association between CAP and MS in CKD patients is mainly caused by BMI and serum triglyceride levels. Obesity is the most significant single risk factor for the development of hepatic steatosis [34], especially excess central or visceral fat [35]. Based on these observations, CAP using transient elastography can be a useful tool to detect MS and central obesity in CKD patients, consistent with previous studies.
Our study has several limitations. First, the assessment of MS was made only at a single time point, and the present study was performed by using a cross-sectional analysis method. For clarifying the clinical impact of CAP on patients with renal impairment, prospective follow-up studies are necessary. Second, the study patients were relatively skewed to stage 1 or 2 CKD; thus, the application of this result to patients with moderate to severe CKD is limited. Finally, since we did not measure muscle strength or skeletal mass, the association between MS and sarcopenic obesity is unclear. Further confirmatory studies to examine the possible mechanisms of the effect of hepatic steatosis on muscle mass and MS development are warranted.
In conclusion, increased CAP measured with transient elastography was significantly associated with an increased risk of MS in CKD patients. These data suggest that hepatic steatosis measured with CAP could be a novel surrogate marker for MS in CKD patients.
Acknowledgments
We thank the Institute of Gastroenterology, Yonsei University College of Medicine for providing data about transient elastography. This work was supported by the Brain Korea 21 Project for Medical Science, Yonsei University, by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MEST) (No. 2014–050098), and by a grant of the Korea Healthcare Technology R&D Project, Ministry of Health and Welfare, Republic of Korea (H14C2003).
Notes
Conflicts of interest
All authors have no conflicts of interest to declare.