| Kidney Res Clin Pract > Volume 41(3); 2022 > Article |
|
Abstract
Background
Methods
Results
Figure 1.
A flow diagram of study population.
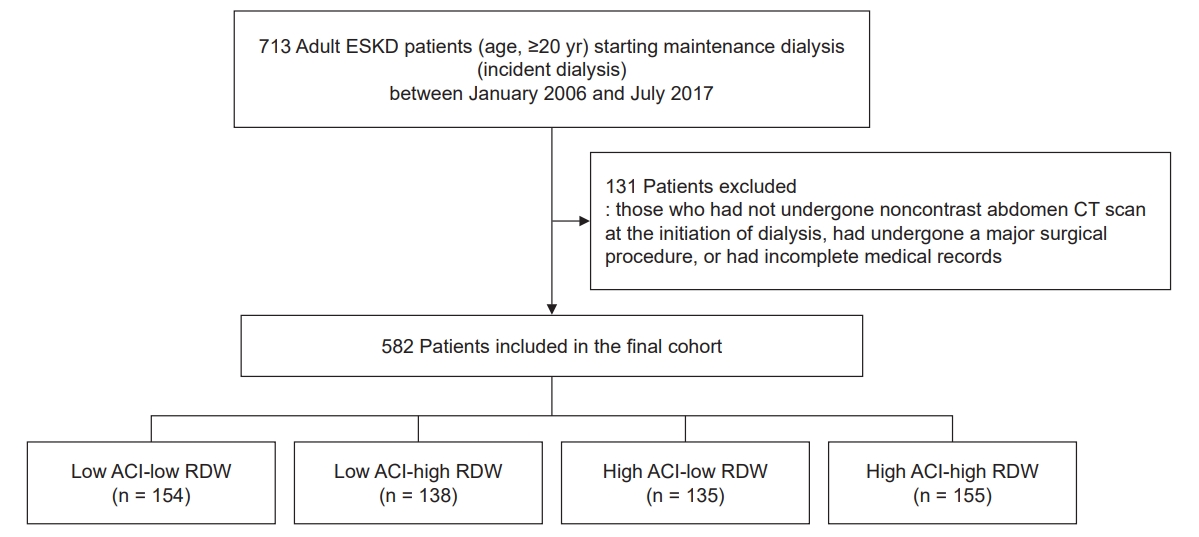
Figure 2.
Comparison of event-free survival rate for all-cause mortality and cardiovascular events according to the ACI and serum RDW value.

Figure 3.
Bivariate correlation analysis of RDW and ACI.

Table 1.
| Variable | Low ACI-low RDW (n = 154) | Low ACI-high RDW (n = 138) | High ACI-low RDW (n = 135) | High ACI-high RDW (n = 155) | p-value |
|---|---|---|---|---|---|
| No. of patients | 154 | 138 | 135 | 155 | |
| Age (yr) | 50.3 ± 12.7 | 53.6 ± 12.5 | 67.0 ± 10.0 | 68.8 ± 10.6 | <0.001 |
| Male sex | 92 (15.8) | 86 (14.8) | 69 (11.9) | 82 (14.1) | 0.17 |
| Body mass index (kg/m2) | 24.0 ± 4.3 | 23.3 ± 3.8 | 24.6 ± 3.8 | 23.2 ± 3.8 | 0.01 |
| Smoking (%)a | 40 (7.0) | 35 (6.1) | 30 (5.2) | 29 (5.0) | 0.36 |
| Diabetes mellitus (%) | 83 (14.3) | 58 (10.0) | 96(16.5) | 96 (16.5) | <0.001 |
| History of CV disease (%) | 19 (3.3) | 14 (2.4) | 41 (7.0) | 41 (7.0) | <0.001 |
| Cause of ESKD (%) | <0.001 | ||||
| Diabetes mellitus | 78 (13.4) | 60 (10.3) | 98 (16.8) | 92 (15.8) | |
| Hypertension | 37 (6.4) | 42 (7.2) | 28 (4.8) | 45 (7.7) | |
| Glomerulonephritis | 17 (2.9) | 12 (2.1) | 4 (0.7) | 4 (0.7) | |
| Others | 22 (3.8) | 24 (4.1) | 5 (0.9) | 14 (2.4) | |
| Dialysis modality | |||||
| Hemodialysis | 99 (17.0) | 99 (17.0) | 104 (17.9) | 115 (19.8) | 0.08 |
| Peritoneal dialysis | 55 (9.5) | 39 (6.7) | 31 (5.3) | 40 (6.9) | |
| eGFR (mL/min/1.73 m2) | 4.2 (2.7–6.7) | 3.9 (2.5–6.4) | 6.1 (4.0–9.8) | 5.3 (3.6–9.4) | <0.001 |
| Creatinine (mg/dL) | 6.9 (5.4–9.4) | 7.2 (5.2–10.3) | 5.5 (4.3–6.9) | 6.1 (4.3–8.4) | <0.001 |
| Urea nitrogen (mg/dL) | 77.5 (58–99) | 84.9 (59–98) | 71.7 (54–89) | 78.2 (61–98) | 0.06 |
| Albumin (g/dL) | 3.6 (3.1–4.0) | 3.4 (2.9–3.9) | 3.4 (2.9–3.9) | 3.3 (2.9–3.6) | 0.003 |
| ALP (U/L) | 101 (77–182) | 108 (82–189) | 119 (82–167) | 102 (78–184) | 0.54 |
| Calcium (mg/dL) | 7.9 (7.2–8.6) | 7.7 (7.2–8.3) | 8.2 (7.6–8.7) | 8.1 (7.6–8.6) | 0.001 |
| Phosphorus (mg/dL) | 5.5 (4.5–7.2) | 5.8 (4.5–7.3) | 5.0 (4.2–6.2) | 5.0 (4.1–6.6) | 0.002 |
| Hemoglobin (g/dL) | 8.9 (7.7–9.9) | 8.8 (7.6–9.7) | 9.3 (8.5–10.2) | 8.9 (7.9–10) | 0.004 |
| RDW (%) | 13.2 (12.7–13.6) | 15.1 (14.5–16.1) | 13.2 (12.9–13.7) | 15.0 (14.6–15.9) | <0.001 |
| Leukocyte count (×103/μL) | 7.5 (5.8–9.8) | 7.4 (5.8–9.9) | 7.4 (5.7–9.7) | 7.8 (6.0–10.2) | 0.71 |
| Platelet count (×103/μL) | 203 (159–261) | 182 (135–247) | 206 (166–258) | 191 (140–255) | 0.04 |
| Total cholesterol (mg/dL) | 170 (137–199) | 158 (125–201) | 153 (121–198) | 157 (126–201) | 0.21 |
| Triglyceride (mg/dL) | 139 (111–203) | 122 (92–172) | 142 (104–181) | 117 (88–154) | <0.001 |
| HDL cholesterol (mg/dL) | 37 (29–48) | 38 (30–49) | 36 (27–45) | 38 (29–47) | 0.36 |
| LDL cholesterol (mg/dL) | 107 (84–134) | 98 (71–128) | 94 (77–127) | 96 (71–125) | 0.08 |
| Intact PTH (pg/mL) | 273 (169–439) | 257 (141–421) | 198 (121–316) | 191 (105–326) | <0.001 |
| hs-CRP (mg/L) | 2.8 (0.9–18.3) | 3.5 (1.1–16.0) | 3.0 (0.9–17.0) | 8.6 (2.3–48.5) | <0.001 |
| Iron (μg/dL) | 57 (40–88) | 57 (36–84) | 56 (39–74) | 47 (26–76) | 0.009 |
| Transferrin saturation (%) | 26.5 (19–41) | 25.6 (17–38) | 25.1 (18–38) | 23.7 (14–36) | 0.1 |
| Ferritin (ng/mL) | 187 (98–326) | 180 (66–339) | 194 (105–339) | 190 (107–434) | 0.499 |
| ACIb | 3.2 ± 5.67 | 3.8 ± 6.06 | 33.9 ± 12.4 | 33.0 ± 12.7 | <0.001 |
| Vitamin B12 (pg/mL) | 756 (512–1,009) | 811 (517–1,087) | 807 (576–1,123) | 845 (640–1,110) | 0.36 |
| Folate (ng/mL) | 7.8 (6–17) | 8.2 (5–19) | 9.9 (6–23) | 13.9 (7–25) | 0.04 |
| ESA | 142 (24.4) | 135 (23.2) | 128 (22.0) | 149 (25.6) | 0.14 |
| Iron replacement | 135 (23.2) | 116 (19.9) | 114 (19.6) | 134 (23.0) | 0.79 |
| Ca-based P-binders | 110 (18.9) | 101 (17.4) | 75 (12.9) | 87 (14.9) | 0.001 |
| Non-Ca-based P-binders | 41 (7.0) | 47 (8.1) | 21 (3.6) | 19 (3.3) | <0.001 |
| Vitamin D analogues | 56 (9.6) | 46 (7.9) | 36 (6.2) | 32 (5.5) | 0.01 |
| RAAS blockers | 118 (20.3) | 95 (16.3) | 97 (16.7) | 107 (18.4) | 0.4 |
| Ca channels-blockers | 108 (18.6) | 102 (17.5) | 97 (16.7) | 124 (21.3) | 0.22 |
| Beta-blockers | 83 (14.3) | 92 (15.8) | 95 (16.3) | 106 (18.2) | 0.01 |
| Statins | 84 (14.4) | 63 (10.8) | 80 (13.7) | 78 (13.4) | 0.13 |
Data are expressed as mean ± standard deviation, number (%), or median (range).
ACI, aortic calcification index; ALP, alkaline phosphatase; Ca, calcium; CV, cardiovascular; eGFR, estimated glomerular filtration rate; ESA, erythropoietin stimulating agent; ESKD, end-stage kidney disease; HDL, high-density lipoprotein; hs-CRP, high sensitivity C-reactive protein; LDL, low-density lipoprotein; P, phosphorus; PTH, parathyroid hormone; RAAS, renin-angiotensin-aldosterone system; RDW, red blood cell distribution width.
Table 2.
Data are expressed as median (interquartile range).
ACI, aortic calcification index; E/A ratio, ratio of peak velocity of early filling (E) to peak velocity of atrial filling (A); E/E’ ratio, ratio of peak velocity of early filling (E) to early diastolic mitral annular early peak velocity (E’); IVST, interventricular septal thickness at end-diastole; LA, left atrium; LV, left ventricle; LVID, left ventricular internal dimension; PW, left ventricular posterior wall; RDW, red blood cell distribution width.
Table 3.
ACI, aortic calcification index; Ca, calcium; CI, confidence interval; CV, cardiovascular; eGFR, estimated glomerular filtration rate; HD, hemodialysis; HR, hazard ratio; hs-CRP, high sensitivity C-reactive protein; P, phosphorus; PD, peritoneal dialysis; RAAS, renin-angiotensin-aldosterone system; RDW, red blood cell distribution width.
Table 4.
ACI, aortic calcification index; Ca, calcium; CI, confidence interval; CV, cardiovascular; E/E’, ratio of mitral peak velocity of early filling (E) to early diastolic mitral annular velocity (E’); eGFR, estimated glomerular filtration rate; HR, hazard ratio; hs-CRP, high sensitivity C-reactive protein; LV, left ventricle; P, phosphorous; RDW, red blood cell distribution width.
References
-
METRICS

- ORCID iDs
-
Da Won Kim

https://orcid.org/0000-0002-0581-6960Munpyo Lee

https://orcid.org/0000-0002-3106-0554Kwon Jae Lee

https://orcid.org/0000-0001-8120-4651Yeon Hee Lee

https://orcid.org/0000-0001-6932-4036Dongryul Kim

https://orcid.org/0000-0002-1322-1887Seok Joon Shin

https://orcid.org/0000-0001-7642-2849Hye Eun Yoon

https://orcid.org/0000-0002-6347-7282 - Related articles
-
The impact of severe depression on the survival of older patients with end-stage kidney disease



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print















