| Kidney Res Clin Pract > Volume 38(2); 2019 > Article |
|
Abstract
Background
P-glycoprotein (P-gp) transports many chemicals that vary greatly in their structure and function. It is normally expressed in renal proximal tubular cells. We hypothesized that P-gp expression influences light chain excretion. Therefore, we investigated whether renal tubular P-gp expression is altered in patients with plasma cell disorders.
Methods
We evaluated renal biopsy specimens from patients with plasma cell disorders (n = 16) and primary focal segmental glomerulosclerosis (the control group, n = 17). Biopsies were stained with an anti-P-gp antibody. Loss of P-gp expression was determined semi-quantitatively. Groups were compared for loss of P-gp expression, and clinical variables.
Results
P-gp expression loss was more severe in patients with plasma cell disorders than it was in those with glomerulonephritis (P = 0.021). In contrast, clinical and histological parameters including serum creatinine, level of urinary protein excretion, and interstitial fibrosis/tubular atrophy grade were not significantly different between the groups. P-gp expression loss increased with age in patients with plasma cell disorders (P = 0.071). This expression loss was not associated with serum creatinine, the level of urinary protein excretion or the interstitial fibrosis/tubular atrophy grade. There was no significant association between the severity of P-gp expression loss with the types and serum levels of light chains, isotypes and serum immunoglobulin levels.
Conclusion
Renal tubular P-gp expression is significantly down-regulated in patients with plasma cell disorders characterized by nephrotic range proteinuria. Additional studies are needed to determine whether reintroduction of renal tubular P-gp expression would mitigate the proximal tubular injury that is caused by free-light chains.
Cubilin and megalin are giant glycoprotein receptors that are abundant on the luminal surface of proximal tubular cells (PTCs) in the kidney [1]. High concentrations of free-light chains (FLCs) in the ultrafiltrate of patients with multiple myeloma can directly injure the PTCs. The cubilinâmegalin complex expressed on PTCs can lead to excessive endocytosis of FLCs. This endocytosis can subsequently trigger apoptotic, proinflammatory, and fibrotic pathways [2]. Blocking light chain endocytosis prevents its nephrotoxic effects on human kidney PTCs [3].
P-glycoprotein (P-gp) is a transporter that excretes xenobiotics, such as cytotoxic compounds, into the gastrointestinal tract, bile, and urine. This transporter is widely expressed in the body. However, certain cell types in the liver, pancreas, kidney, colon, and jejunum have specific and dominant localization of P-gp [4]. In the kidney, P-gp is predominantly expressed on the apical surface of proximal tubule epithelium.
One of the most interesting characteristics of P-gp/multidrug resistance protein 1 (MDR1) is that its many substrates vary greatly in their structure and function. These substrates range from small molecules, including organic cations, carbohydrates, amino acids, and some antibiotics to macromolecules, such as polysaccharides and proteins [5]. Therefore, P-gp modulation is of interest for various purposes. P-gp is also an amyloid-beta efflux pump. Lam et al [6] described how selected members of the ATP-binding cassette (ABC) superfamily of transporters are responsible for the energy-dependent efflux of a variety of lipophilic and amphipathic molecules from cells. This group hypothesized that an ABC transporter might be responsible for cellular amyloid-beta release. They were able to directly measure the transport of amyloid-beta peptides across the plasma membranes of P-gp enriched vesicles, and showed that this phenomenon was both ATP- and P-gp-dependent. In another study, Lee et al [7] showed that colistin induces nephrotoxicity by suppressing P-gp. In addition, P-gp induction ameliorates colistin-induced nephrotoxicity by decreasing apoptosis.
The physicochemical properties of immunoglobulins (Igs), and their regions may differ. The properties of the involved Ig molecules appear to be the primary pathologic determinants of the specific pattern of tissue injury. Although amyloid light-chain (AL) amyloidosis is more commonly caused by lambda (Îŧ) light chain, kappa (Κ) chain isotype restriction is observed in ~80% of cases with light-chain deposition disease. Particular types of light chains may have more hydrophobic properties, and therefore more easily precipitate in tissues compared to other light chain types [8,9].
Based on these observations, we hypothesized modulating P-gp expression would alleviate the tubular damage caused by light chains that are produced by monoclonal B-cells in plasma cell disorders. Because many substrates of P-gp are hydrophobic [10], and some types of FLCs are formed by hydrophobic residues, we suspected that light chains might be a substrate of P-gp. We studied whether renal tubular P-gp expression is altered in patients with plasma cell disorders and primary focal segmental glomerulosclerosis (FSGS) who have comparable proteinuria levels.
Clinical data were collected retrospectively from the medical records. Individuals with a kidney biopsy at the time of the diagnosis of a plasma cell disorder were included. The control group included patients with FSGS to compare the effects of various types of proteinuria on renal P-gp expression (proteinuria caused by excessive light-chain production versus proteinuria of glomerular origin). Participants were above 18 years old. Exclusion criteria included the presence of diabetes mellitus, exposure to any drug that regulates P-gp expression and function (including immunosuppressive drugs). The laboratory studies were collected immediately prior to the renal biopsy. Serum biochemistry results, urinary protein excretion and full blood counts were recorded. Informed consent was gathered from all patients or their relatives for the use of medical data. This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Dokuz EylÞl University (No. 2017/22-04). Informed consent was obtained from all individual participants.
Renal biopsy specimens were evaluated with hematoxylin-eosin, Massonâs trichrome, periodic acid schiff, and methenamine silver staining. The specimens were also stained with antibodies against IgG, IgA, IgM, complement 3c, C1q, kappa and lambda for immunofluorescence. Congo red staining was performed to evaluate for amyloid deposition. If Congo red staining was positive, amyloid-A was evaluated by immunohistochemistry. The tissue sections were examined by two independent pathologists. The minimum required glomeruli for light microscopy and immunofluorescence microscopy were 10 and 3, respectively. The diagnosis and interstitial fibrosis/tubular atrophy (IFTA) scores were retrieved from previous pathology reports, which were scored as follows: normal = 0 point, atrophy in < 10% of tubules placed in cortical area; mild IFTA = 1 point, atrophy in 10% to 24% of tubuli in the cortical area; moderate IFTA = 2 points, atrophy in 25% to 50% of tubuli in the cortical area; severe IFTA = 3 points, atrophy in > 50% of tubuli in the cortical area [11].
The presence of a clonal plasma cell disorder was assessed by serum and urine immunofixation electrophoresis and bone marrow biopsy. The percentage of plasma cells in the bone marrow was estimated. The clonality was evaluated with standard immunohistochemical staining for kappa and lambda light chains. Congo red staining was performed to identify amyloid depositions in the marrow biopsy specimens. A diagnosis of AL amyloidosis required a biopsy with Congo red staining (for amyloid fibrils), and evidence of a clonal plasma cell dyscrasia, per standard criteria [12]. Multiple myeloma was also diagnosed based on the standard criteria [13].
Sections from the paraffin-embedded blocks (of the renal biopsies) were kept in 950C CC1 (cell conditioning 1; Ventana Medical Systems, Tuscon, USA) solution for 52 minutes, and then incubated in prediluted primary monoclonal anti-P-gp antibody (dilution, 1/500; EPR10364-57; Abcam Inc., Cambridge, USA). The stainings were performed in the Benchmark Ultra (Ventana Medical Systems) device using the Ultraview Universal DAB Detection kit (Ventana Medical Systems). The kidney tissue was used as a positive control. P-gp expression was assessed at the glomeruli and the tubular epithelial cells. In most cases, the glomeruli were negative. If any positivitiy was identified, the number of positive glomeruli was noted. Expression was scored semi quantitatively according to the intensity and distribution. The classification was as follows: normal = 0 point, P-gp expression loss in < 10% of tubules placed in the cortical area; mild loss = 1 point, P-gp expression loss in 10% to 24% of tubuli in the cortical area; moderate loss = 2 points, P-gp expression loss in 25% to 50% of tubuli in the cortical area; severe loss = 3 points, P-gp expression loss in > 50% of tubuli in the cortical area; as modified from a previous study by Koziolek et al [14]. We used these cut-off levels because the IFTA grade is also scored using a similar classification while evaluating the same anatomic location for P-gp expression.
Variable distributions were assessed by the KolmogorovâSmirnov test. Quantitative variables are expressed as means Âą standard deviations if normally distributed, or as medians (ranges) with the interquartile range otherwise. Qualitative variables are expressed as proportions. The groups were compared for means using the t test if variables were normally distributed. The MannâWhitney U test was used to compare means if the variables were not normally distributed. For comparisons between proportions, chi-squared tests or Fisherâs exact test were used, as appropriate. Statistical analysis was performed using IBM SPSS Statistics ver. 22.0 (IBM Corp., Armonk, USA). P values less than 0.05 were considered statistically significant.
Five patients from the plasma cell disorder group and 7 patients with FSGS were excluded because of insufficient biopsy specimens for immunohistochemical analysis. There were 16 patients in the plasma cell disorders group. All of the patients in this group had a diagnosis of primary amyloidosis and/or multiple myeloma. The control group included 17 patients with FSGS.
P-gp expression was dominant in the renal proximal tubules in all biopsies (Fig. 1). Expression loss was significantly more severe in patients with plasma cell disorders than it was in patients with glomerulonephritis. In contrast, the clinical and histological parameters, including serum creatinine, level of urinary protein excretion, and IFTA grade, were not significantly different between the groups (Table 1). Distribution of patients according to severity of P-glycoprotein expression loss is presented in Fig. 2.
When all patients were included in the same analysis and patients were classified as normal-mild P-gp expression loss and moderate-severe P-gp expression loss, age was the only parameter that was significantly different between the groups (P = 0.033). In contrast, the serum creatinine, urinary protein excretion levels, and IFTA grade were similar between the two groups.
Of patients with plasma cell disorders, the serum creatinine, urinary protein excretion levels and and IFTA grade were not significantly different between the two groups with various grades of P-gp expression loss (Table 2). Although the expression loss increased with age (Fig. 3), this was not statistically significant. The P-gp expression loss was not associated with the serum creatinine, level of urinary protein excretion, or IFTA grade. There was no significant association between the severity of P-gp expression loss with the types and serum levels of light chains, isotypes, and serum levels of Igs.
When patients with plasma cell disorders were grouped according to the severity of the loss of P-gp expression, there was no significant difference in the clinical parameters between the groups. Acute kidney injury was the most frequent indication for kidney biopsy in patients with plasma cell disorders (comprising 68.8% of patients).
In patients with plasma cell disorders, 3 had primary amyloidosis, 6 had multiple myeloma and 7 had both. When patients were evaluated for monoclonality, seven of them had lambda, 2 IgG-lambda, 3 kappa, 3 IgG-kappa, and 1 IgA-kappa monoclonal gammopathy. Cast nephropathy was evident in the kidney biopsies of 7 patients. P-gp expression was not significantly different between patients with primary amyloidosis and multiple myeloma. There was no significant association between the severity of P-gp expression loss between different types and serum levels of light chains, and isotypes and serum levels of Igs. The P-gp expression was not different between patients with or without cast nephropathy. The clinical and histopathological properties of patients with plasma cell disorders are given in Table 3.
In this study, we showed that renal tubular P-gp expression is more significantly downregulated in patients with plasma cell disorders than it is in FSGS patients with similar proteinuria levels. The control group included patients with FSGS so that we could analyze the effect of intratubular light chain burden compared to that of proteinuria of glomerular origin. Interestingly, the level of proteinuria in patients plasma cell disorders was not significantly associated with altered P-gp expression. We did not identify an association between any specific type of Ig fragment and P-gp expression.
The clinical variables were not significantly different between the patient groups with variable P-gp expression (Table 2). It is not clear why the P-gp expression is altered in some patients with plasma cell disorders, but not in others. The substrates of P-gp are cationic and hydrophobic molecules. Light chains differ in their physicochemical structure. Therefore, more hydrophobic or more cationic light chains may be more frequently associated with altered P-gp expression than are others. Unfortunately, our sample size was too low to demonstrate the effect of a particular light chain on P-gp expression pattern.
Since the P-gp is expressed in the apical border of the proximal tubular epithelium, tubular atrophy probably affects its expression levels. However, tubular atrophy itself is not an absolute obstacle for P-gp expression. Koziolek et al [14] found that severely damaged tubules, such as those in acute tubular necrosis, were still able to upregulate P-gp after cyclosporine-A exposure. In addition, patients with acute vascular or chronic allograft rejection (who displayed a similar degree of long-term renal dysfunction) also produced increased P-gp expression. Therefore, although tubular atrophy may affect the tubular P-gp expression, tubular atrophy itself is not an absolute predictor of altered expression. Instead, the loss of P-gp expression itself causes tubular injury [15]. Therefore, an increase in P-gp expression seems to be a compensatory way to reduce tubular injury [16]. In a study, endotoxemia caused by lipopolysaccharide administration lead to proximal tubular injury [15]. In contrast, lipopolysaccharide administration led to an increase in P-gp expression. Therefore, although the surviving brush borders may alter expression, tubular injury itself may inversely increase P-gp expression. The mean IFTA scores were higher in plasma cell disorders than they were in patients with FSGS in our paper. However, the IFTA scores were similar between patient groups with different grades of P-gp expression (Table 2). We presented examples that show severely reduced P-gp expression despite mild tubular atrophy, or only mild expression loss despite severe tubular atrophy. Since P-gp is mainly expressed in the brush borders of the proximal tubules, tubular atrophy may alter P-gp expression. However, tubular injury may also inversely increase P-gp expression. In our opinion, the cause of tubular injury likely influences the nature of P-gp expression change. Regardless, this issue requires further investigation.
The effect of renal failure on P-gp expression is not clear. An animal study found that P-gp expression was unchanged in a rat model of chronic renal failure [17]. Lee et al [7] found that colistin itself suppressed the P-gp expression, and lead to nephrotoxicity in PTC culture. Following dexamethasone administration, P-gp expression increased and nephrotoxicity was alleviated. Huls et al [16] found that P-gp was upregulated after acute renal injury secondary to ishchemia-reperfusion damage. Based on these findings, the group suggested that the P-gp upregulation after acute kidney injury reflected the transporterâs role in the renal regeneration process. The group added that P-gp upregulation may serve as a protective function by pumping out toxins and harmful products of oxidative stress. One group caused renal injury by culturing proximal tubule cells in cadmium; they found that P-gp expression was upregulated after renal injury, and suggested that P-gp may be protective against cadmium- and reactive oxygen species-induced apoptosis [18]. An experimental study performed by Huang et al [19] showed that P-gp expression (in the kidney) was increased in rats with glycerol-induced acute renal injury. However, the function of P-gp, which was evaluated by measuring the renal secretory clearance of rhodamine 123, was suppressed. Although increased creatinine may be a confounder, the groups were not significantly different in terms of kidney function.
Among patients with plasma cell disorders, P-gp expression decreased with age. Although patients with plasma cell disorders were older on average, there was not a significant difference in age between the two groups. It is not clear how P-gp expression is modulated during aging. Warrington and colleagues studied P-gp expression and function in rats [20]. The authors reported that although lymphocytic and hepatic P-gp expression increased with age, the renal P-gp content decreased with age. Another study showed increased expression of P-gp at both the protein and the mRNA levels in aging lymphocytes [21]. Therefore, the role of aging on P-gp expression appears to be complex, and that in the human kidney itself is unknown.
The P-gp expression was primarily demonstrated in the brush border of the proximal tubule epithelium. This was first demonstrated by Thiebaut et al [4], who stated that no P-gp expression was established in any other cells in the kidney. Multiple subsequent papers supported the finding that P-gp is primarily found in proximal tubules [22â24], although some have described P-gp expression in the distal tubules [14]. Thiebaut et al [4] concluded that normal P-gp expression in the body is almost always identified on the apical sides of organ systems, except in the adrenal glands. It is also mainly distributed in the apical membranes of proximal tubular epithelium of the kidney. Therefore, although some papers demonstrate P-gp expression in various parts of the nephron, the proximal tubules should be considered the primary site of expression.
P-gp reduces the exposure of cancer cells to chemotherapeutics leading to chemotherapeutic resistance. Therefore, there has been much interest in its expression. In nephrology, P-gp expression is studied particularly in the renal transplantation era because pharmacokinetics of calcineurin inhibitors is altered by P-gp [25]. This alteration may cause serious drug-drug interactions, transplant rejection or calcineurin-inhibitor toxicity. Although we found that P-gp expression is downregulated in patients with plasma cell disorders, the clinical significance of this expression is unknown.
FLC burden is an important determinant of renal injury in patients with multiple myeloma and primary amyloidosis. Therapies that eliminate monoclonal plasma cells ameliorate the organ toxicity of FLCs [26]. Data from our preliminary study suggest that renal tubular P-gp expression is markedly downregulated in patients with multiple myeloma and primary amyloidosis with nephrotic-range proteinuria. It is unknown whether inducing P-gp expression increases the efflux of light chains from the tubular epithelial cells. The substrates of P-gp differ greatly by their function and structure. Typical P-gp substrates are lipophilic or amphipathic large molecules (molecular weight > 400 Da) [27]. Therefore, light chains with various structures could be secreted by the P-gp system from renal tubular epithelial cells. Although modulating P-gp may alleviate proximal tubular injury, it is unknown whether this aggravates cast formation by increased delivery of light chains to the distal tubules. We could not demonstrate the effects of particular urinary protein content (albumin, globulin, light chain) on P-gp expression directly, because the specific types of proteins are not routinely quantified in urine. This issue is of significant importance, and must be clarified in future studies.
Our sample size was small. Therefore, our results should be substantiated by larger studies. The possible effects of particular light chain types might be observed if larger numbers of patients were included. The evaluation and grading of expression were made semiquantitatively, which may have led to overestimation or underestimation of the expression. Although P-gp expression has been quantified using western blot analysis in several prospective experimental researches [28], there are very few studies in which renal tubular P-gp expression was evaluated retrospectively from previous biopsy specimens. We used a similar method to that of Koziolek et al [14], except that we used different cut-off levels to determine P-gp expression. We used the same cut-off levels that are applied to IFTA scoring, since IFTA is also determined by examining a similar anatomical part of the kidney.
In conclusion, renal tubular P-gp expression is down-regulated in patients with primary amyloidosis and/or multiple myeloma with nephrotic-range proteinuria compared to that of patients with primary FSGS (with comparable proteinuria). Further studies are needed to confirm our results, and to determine whether FLCs in renal tubuli are important for regulating P-gp expression. If so, P-gp induction may alleviate renal tubular injury caused by FLC.
Acknowledgments
We thank Professor HÞlya Ellidokuz for her contributions to the statistical analysis. Our study was supported by the Dokuz EylÞl University Coordinating Subcommittee for Scientific Research Projects (DEU BAP).
Notes
Authorsâ contributions
Cihan Heybeli, Mehmet Asi Oktan, and Hayri Ustun Arda collected the data and wrote the manuscript. Cihan Heybeli, Caner Cavdar, Sulen Sarioglu, and Taner Camsari participated in the study design and performed the statistical analysis. Cihan Heybeli, Mehmet Asi Oktan, Sulen Sarioglu, and Caner Cavdar participated in the conception, analysis, and interpretation of data. Serkan Yildiz, Mehtat Unlu, Aykut Sifil, Ali Celik, and Taner Camsari provided intellectual content of critical importance to the work and technical support. Sulen Sarioglu and Caner Cavdar participated in the study design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Figure 1
P-glycoprotein (P-gp) expression on renal biopsy (100Ã)
(A) Normal P-gp expression in a patient with focal segmental glomerulosclerosis. Mild interstitial fibrosis/tubular atrophy (IFTA) was noted in the biopsy report. (B) A kidney section of a patient with multiple myeloma who had mild P-gp expression loss (expression loss in 10â24% of tubules in cortical area). Although there is no clear atrophy in the central tubuli, no P-gp expression is seen in this area. (C) P-gp expression in a kidney of a patient with focal segmental glomerulosclerosis is presented. Although there is severe tubular atrophy, P-gp expression loss was regarded as mild. (D) Severe P-gp expression loss (expression loss in > 50% tubules in cortical area) in a patient with primary amyloidosis + multiple myeloma is seen. This biopsy was reported to show mild IFTA.
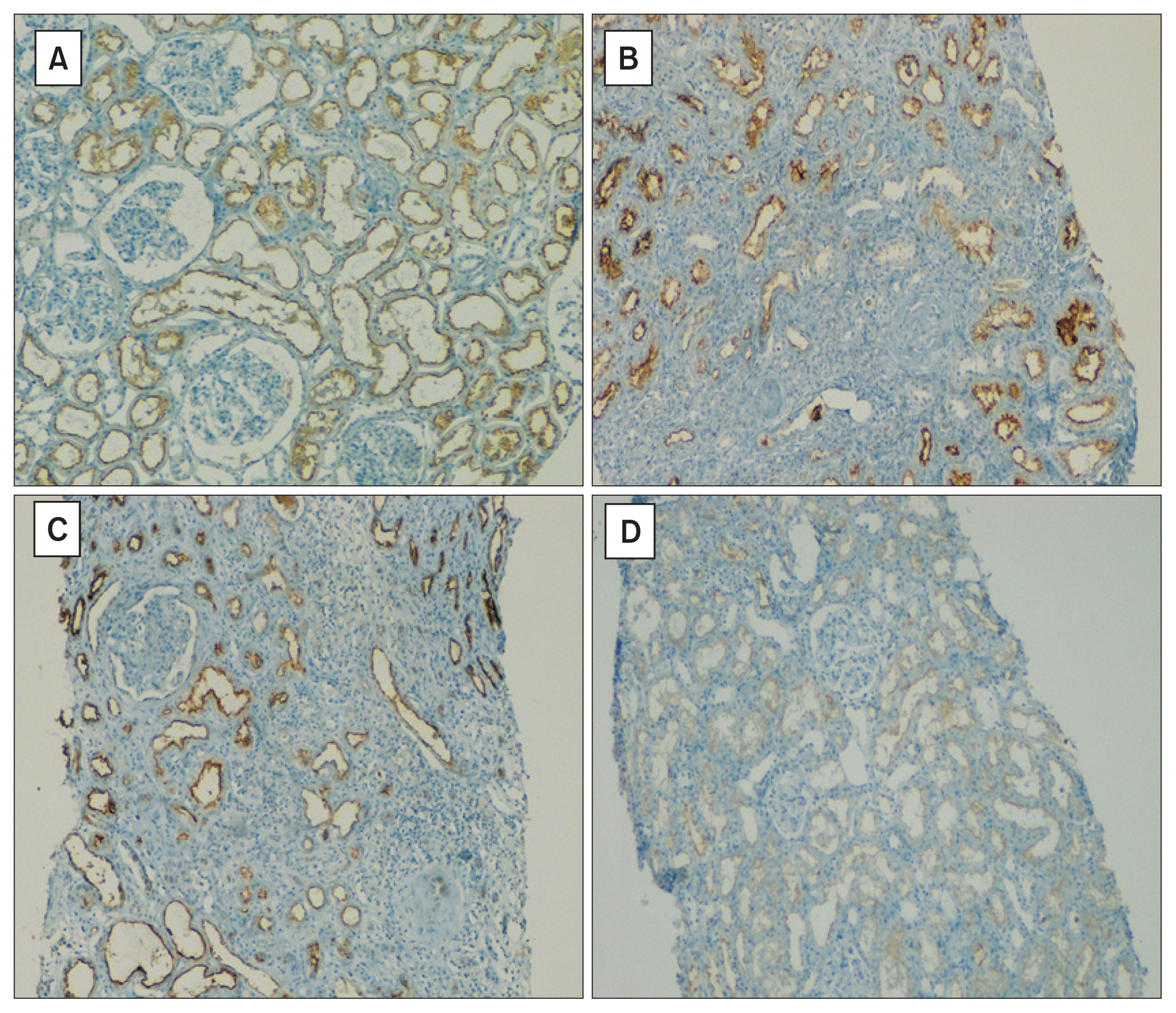
Figure 2
Distribution of patients according to the severity of P-glycoprotein (P-gp) expression loss.
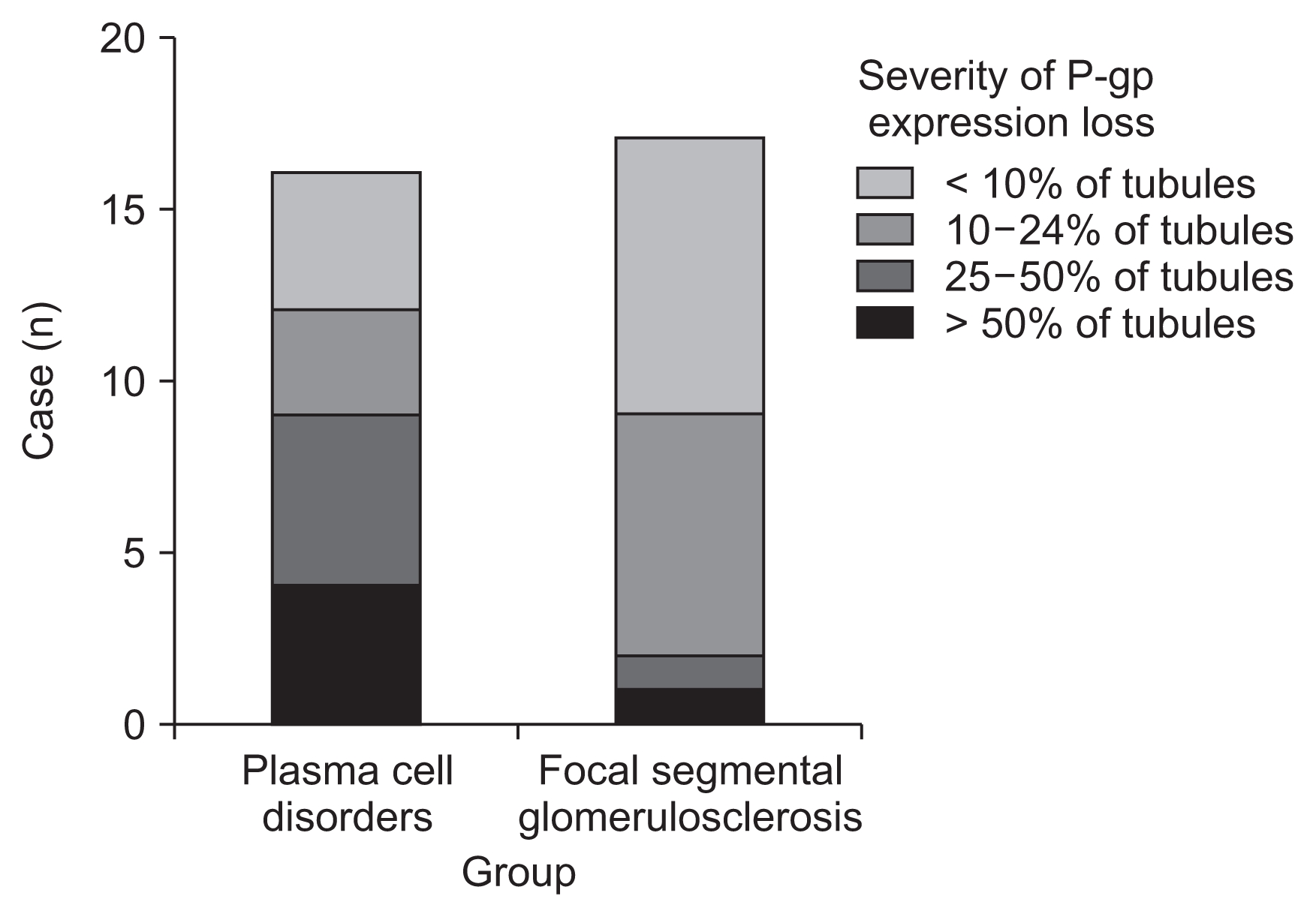
Figure 3
Distribution of patients in different age groups according to the severity of P-glycoprotein (P-gp) expression loss.
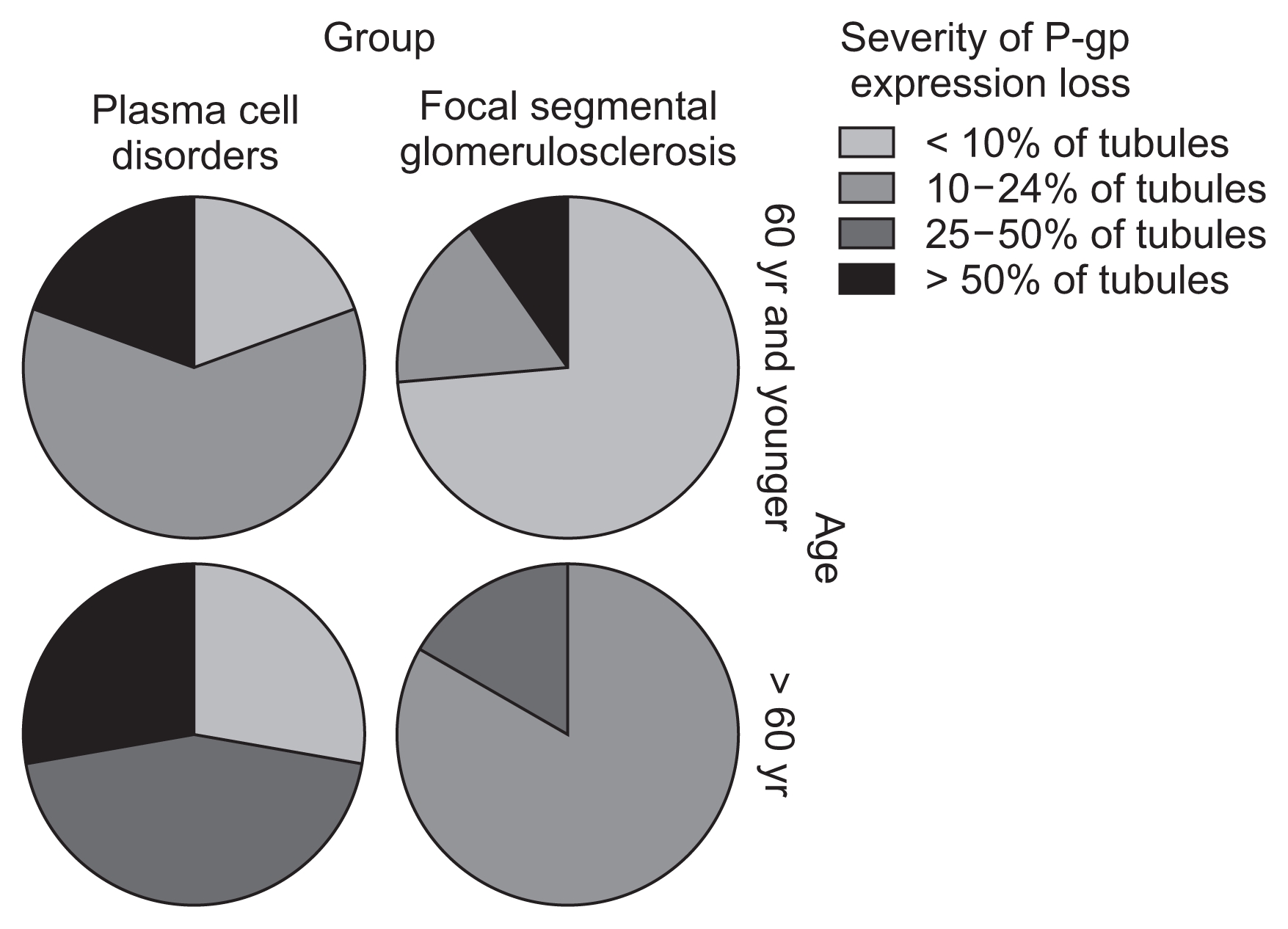
Table 1
Comparative analysis of the clinical and histopathological findings of patients with plasma cell disorders and FSGS
| Variable | Plasma cell disorder (n = 16) | FSGS (n = 17) | P value |
|---|---|---|---|
| Age (yr) | 60.8 Âą 8.1 | 56.5 Âą 14.3 | 0.533 |
| Sex, female | 6 (37.5) | 7 (41.2) | 0.829 |
| Serum creatinine (mg/dL) | 2.35 Âą 0.96 | 1.94 Âą 1.14 | 0.382 |
| Proteinuria (g/day) | 7.4 Âą 3.7 | 7.4 Âą 5.2 | 0.581 |
| Hemoglobin (g/dL) | 11.0 Âą 1.9 | 12.0 Âą 2.4 | 0.458 |
| Albumin (g/dL) | 3.2 Âą 0.9 | 3.1 Âą 0.7 | 0.960 |
| IFTA (%) | NA | ||
| â< 10 | 0 | 1 | |
| â10â24 | 4 | 8 | |
| â25â50 | 6 | 1 | |
| â> 50 | 6 | 7 | |
| IFTA scorea | 2.1 Âą 0.8 | 1.9 Âą 1.1 | 0.444 |
| P-gp expression loss (%) | NA | ||
| â< 10 | 4 | 8 | |
| â10â24 | 3 | 7 | |
| â25â50 | 5 | 1 | |
| â> 50 | 4 | 1 | |
| P-gp expression loss in âĨ 25% of cortical tubules | 9 (56.3) | 3 (17.6) | 0.021 |
| Score of P-gp expression lossa (point) | 1.56 Âą 1.15 | 0.71 Âą 0.85 | 0.037 |
Table 2
Comparative analysis of clinical and histopathological findings of patients with plasma cell disorders according to the percent of P-glycoprotein loss in cortical tubules
| Variable | Loss of expression | P value | |
|---|---|---|---|
|
|
|||
| Normal or mildly (n = 7) | Moderate or severe (n = 9) | ||
| Age (yr) | 56.1 Âą 9.4 | 64.4 Âą 4.6 | 0.071 |
| Sex, female | 4 (57.1) | 2 (22.2) | 0.302 |
| Indication for renal biopsy | NA | ||
| âAKI | 6 | 5 | |
| âNS | 1 | 2 | |
| âCKD | 0 | 2 | |
| Serum creatinine (mg/dL) | 2.34 Âą 0.77 | 2.35 Âą 1.14 | 0.918 |
| Delta serum creatininea (mg/dL) | 1.14 Âą 0.6 | 1.7 Âą 1.04 | 0.686 |
| Proteinuria (g/day) | 8.2 Âą 5.0 | 6.7 Âą 2.4 | 0.681 |
| Hemoglobin (g/dL) | 11.1 Âą 1.9 | 11.0 Âą 1.9 | 0.950 |
| Albumin (g/dL) | 3.4 Âą 0.8 | 3.0 Âą 0.96 | 0.366 |
| IFTA (%) | NA | ||
| â< 10 | 0 | 0 | |
| â10â24 | 1 | 3 | |
| â25â50 | 4 | 2 | |
| â> 50 | 2 | 4 | |
| IFTA scoreb | 2.14 Âą 0.69 | 2.11 Âą 0.93 | 1.000 |
| Final diagnosis | NA | ||
| âAmyloidosis | 1 | 2 | |
| âMM + amyloidosis | 4 | 3 | |
| âMM | 2 | 4 | |
Table 3
Clinical and histopathological characteristics of patients with plasma cell disorders
References
1. Klassen RB, Allen PL, Batuman V, Crenshaw K, Hammond TG. Light chains are a ligand for megalin. J Appl Physiol (1985) 2005;98:257â263.


2. Hutchison CA, Batuman V, Behrens J, et al. International Kidney and Monoclonal Gammopathy Research Group. The pathogenesis and diagnosis of acute kidney injury in multiple myeloma. Nat Rev Nephrol 2011;8:43â51.



3. Li M, Balamuthusamy S, Simon EE, Batuman V. Silencing megalin and cubilin genes inhibits myeloma light chain endocytosis and ameliorates toxicity in human renal proximal tubule epithelial cells. Am J Physiol Renal Physiol 2008;295:F82âF90.


4. Thiebaut F, Tsuruo T, Hamada H, Gottesman MM, Pastan I, Willingham MC. Cellular localization of the multidrug-resistance gene product P-glycoprotein in normal human tissues. Proc Natl Acad Sci U S A 1987;84:7735â7738.



5. Zhou SF. Structure, function and regulation of P-glycoprotein and its clinical relevance in drug disposition. Xenobiotica 2008;38:802â832.


6. Lam FC, Liu R, Lu P, et al. Îē-Amyloid efflux mediated by p-glycoprotein. J Neurochem 2001;76:1121â1128.


7. Lee SH, Kim JS, Ravichandran K, Gil HW, Song HY, Hong SY. P-glycoprotein induction ameliorates colistin induced nephrotoxicity in cultured human proximal tubular cells. PLoS One 2015;10:e0136075



8. Paueksakon P, Fogo AB. More light shed on light chains. Nephrol Dial Transplant 2014;29:1799â1801.


9. DeKosky BJ, Lungu OI, Park D, et al. Large-scale sequence and structural comparisons of human naive and antigen-experienced antibody repertoires. Proc Natl Acad Sci U S A 2016;113:E2636âE2645.



10. Sharom FJ, Liu R, Romsicki Y, Lu P. Insights into the structure and substrate interactions of the P-glycoprotein multidrug transporter from spectroscopic studies. Biochim Biophys Acta 1999;1461:327â345.


11. Sethi S, DâAgati VD, Nast CC, et al. A proposal for standardized grading of chronic changes in native kidney biopsy specimens. Kidney Int 2017;91:787â789.


12. Gertz MA, Comenzo R, Falk RH, et al. Definition of organ involvement and treatment response in immunoglobulin light chain amyloidosis (AL): a consensus opinion from the 10th International Symposium on Amyloid and Amyloidosis, Tours, France, 18â22 April 2004. Am J Hematol 2005;79:319â328.


13. Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014;15:e538âe548.


14. Koziolek MJ, Riess R, Geiger H, ThÃĐvenod F, Hauser IA. Expression of multidrug resistance P-glycoprotein in kidney allografts from cyclosporine A-treated patients. Kidney Int 2001;60:156â166.


15. Heemskerk S, van Koppen A, van den Broek L, et al. Nitric oxide differentially regulates renal ATP-binding cassette transporters during endotoxemia. Pflugers Arch 2007;454:321â334.



16. Huls M, Kramers C, Levtchenko EN, et al. P-glycoprotein-deficient mice have proximal tubule dysfunction but are protected against ischemic renal injury. Kidney Int 2007;72:1233â1241.


17. Laouari D, Yang R, Veau C, Blanke I, Friedlander G. Two apical multidrug transporters, P-gp and MRP2, are differently altered in chronic renal failure. Am J Physiol Renal Physiol 2001;280:F636âF645.


18. ThÃĐvenod F, Friedmann JM, Katsen AD, Hauser IA. Up-regulation of multidrug resistance P-glycoprotein via nuclear factor-kappaB activation protects kidney proximal tubule cells from cadmium- and reactive oxygen species-induced apoptosis. J Biol Chem 2000;275:1887â1896.


19. Huang ZH, Murakami T, Okochi A, Yumoto R, Nagai J, Takano M. Expression and function of P-glycoprotein in rats with glycerol-induced acute renal failure. Eur J Pharmacol 2000;406:453â460.


20. Warrington JS, Greenblatt DJ, von Moltke LL. The effect of age on P-glycoprotein expression and function in the Fischer-344 rat. J Pharmacol Exp Ther 2004;309:730â736.


21. Aggarwal S, Tsuruo T, Gupta S. Altered expression and function of P-glycoprotein (170 kDa), encoded by the MDR 1 gene, in T cell subsets from aging humans. J Clin Immunol 1997;17:448â454.

22. Wessler JD, Grip LT, Mendell J, Giugliano RP. The P-glycoprotein transport system and cardiovascular drugs. J Am Coll Cardiol 2013;61:2495â2502.


23. Heemskerk S, Peters JG, Louisse J, Sagar S, Russel FG, Masereeuw R. Regulation of p-glycoprotein in renal proximal tubule epithelial cells by LPS and TNF-alpha. J Biomed Biotechnol 2010 2010:525180.
24. Tramonti G, Xie P, Wallner EI, Danesh FR, Kanwar YS. Expression and functional characteristics of tubular transporters: P-glycoprotein, PEPT1, and PEPT2 in renal mass reduction and diabetes. Am J Physiol Renal Physiol 2006;291:F972âF980.


25. Naesens M, Lerut E, de Jonge H, Van Damme B, Vanrenterghem Y, Kuypers DR. Donor age and renal P-glycoprotein expression associate with chronic histological damage in renal allografts. J Am Soc Nephrol 2009;20:2468â2480.



26. Palladini G, Kyle RA, Larson DR, Therneau TM, Merlini G, Gertz MA. Multicentre versus single centre approach to rare diseases: the model of systemic light chain amyloidosis. Amyloid 2005;12:120â126.


- TOOLS



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print
















